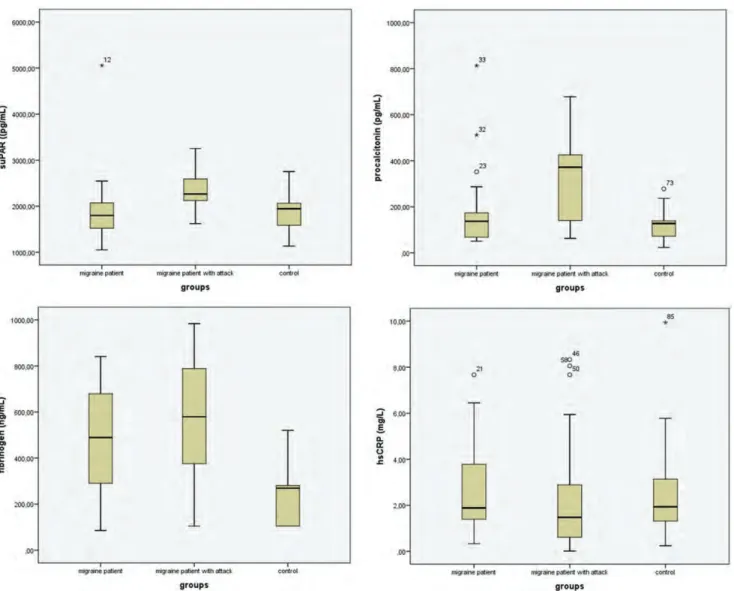
The relationship between levels of plasma-soluble urokinase plasminogen activator receptor (suPAR) and presence of migraine attack and aura
Tam metin
Şekil


Benzer Belgeler
While erythrocyte sedimentation rate had a positive correlation with mean platelet volume, we found no correlation between NLR and other parameters of disease activity, PTX3,
The patients with available results of both remission and attack periods were included in the study.Since the distribution of all continuous variables used in the study as a
雲嘉校友分會 9/13 校友大會回顧 (編輯部整理) 北醫牙醫學系校友會雲嘉分會第一屆第二次會員大會,於 9/13 舉行,前排中著灰色領帶者,為雲嘉分會長 謝炎銘醫師/雲嘉分會提供
permeability, swelling ratio, adhesion test withHEMA, and also the biocompatibility of PDCM, as it is produced following a specific laboratory protocal.. The results of
İki yüz yıl önce İsianbul’a göçmüş Üs- küplü bir ailenin artık çoktan İstanbullu olan çocuğu Muhtar Kargılı, Kanaat Lo kantasını Ü sküdar’da açtığında
Basın tarihi açısından ise partinin yayın organları olarak ortaya çıkan Yurdda Kalkınma, Tez Kalkınma ve Öz Kalkınma gazeteleri, Cumhuriyet döneminin ilk muhalif parti
Tarde’ın kamusal alan modeli; kamunun ortaya çıkışı, karşılıklı konuşmaların teşvik edilmesi, kamuoyunun dile getirilmesi ve oluşumu, bireylerin toplumsal eylemler
E¤itim alan›nda yap›lan araflt›rmalardan çok az› üni- versitelerde yap›lm›fl; Björkqvist, Österman ve Hjelt-Bäck (1994) ve Lewis (2004) ö¤retim