Does vitamin D affect diabetic neuropathic pain and balance?
Tam metin
Şekil

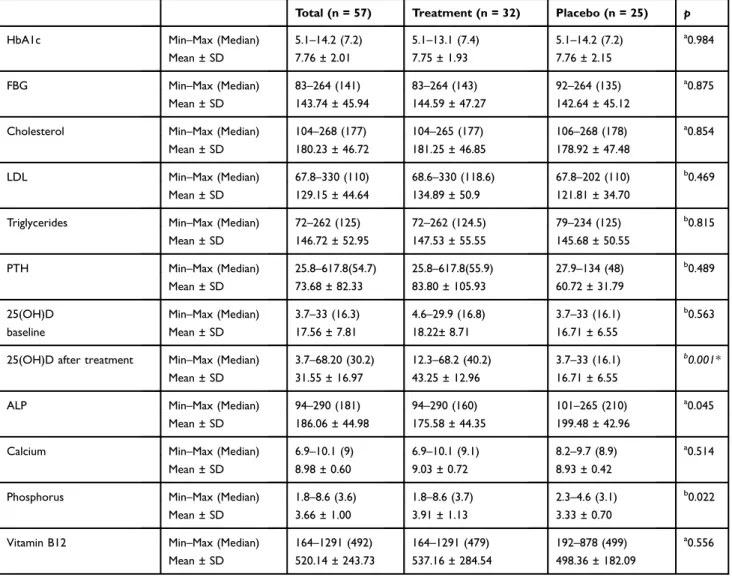
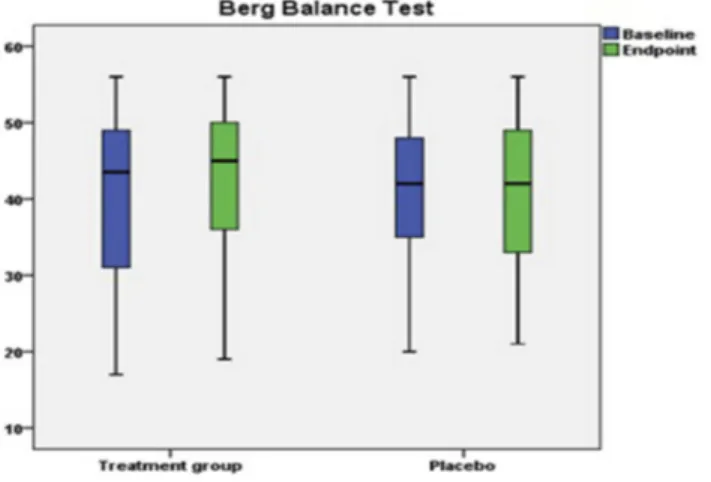
Benzer Belgeler
In this study, the purified destruxin B (DB) was tested for its in vitro and in vivo anti-tumor activities by using L5178Y lymphoma cells which was induced by methylcholanthrene
c Assistant professor, Dept. In recent times, Sign language is omnipresent. However, there exists a challenge for people who do not know sign language, to communicate with people
In an- other study conducted on postmenopausal women who are older than 50 years, women with 25OHD <30 ng/ml were shown to have lower grip strength and lower extremity
In this study, the oral symptoms of a patient admitted to our clinic with dysphagia, periodontal problems and mouth opening limitation ave been presented..
kabukla altüst olmuş bir hâlde ve bu şekilde göçmenin kaderini onaylamadığını gösteren bir şekilde sunulur. Bu kabuk Ayşe’nin hayata karşı duyarsız olarak
[r]
• Exposure to sunlight for 10-15 min 2-3 times a week is sufficient for the body's vitamin D needs.. • However, due to environmental factors
• İnce barsaklarda Ca ve fosfat absorbsiyonunda artışa neden olur.. Vitamin D fonksiyonu.. 1) Vücutta Ca ve fosfat tutulmasını sağlayıp bu minerallerin kan