Comparison of soil fungi flora in burnt and unburnt forest soils in the vicinity of Kargıcak (Alanya, Turkey)
Tam metin
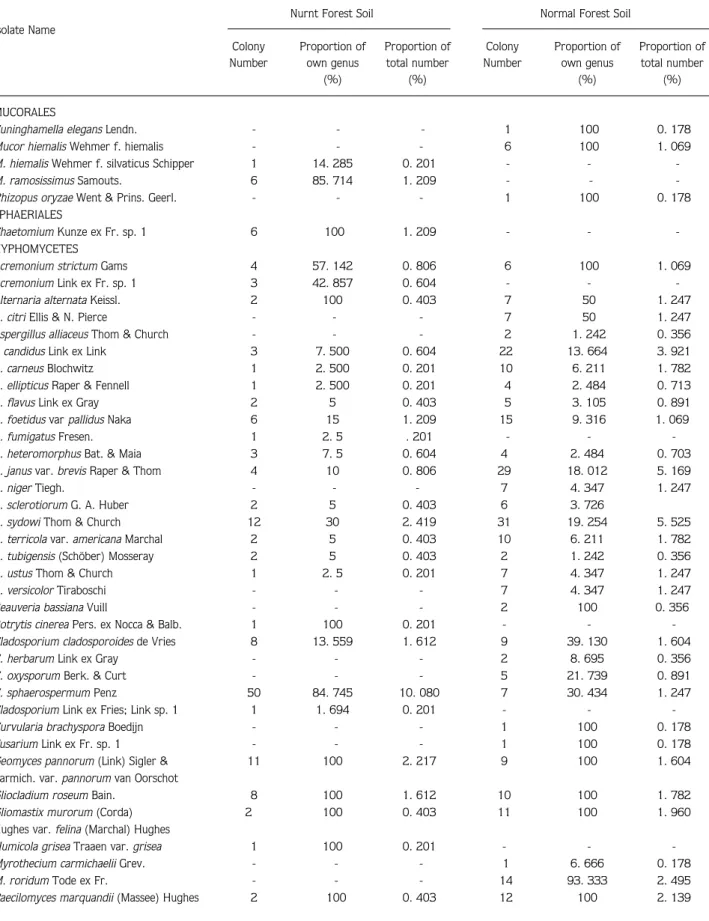
Şekil


Benzer Belgeler
This equation shows that the existence of quantised energy levels depends on the barrier potential V o (energy depth of the single RQW), the length a of the AR as well as
Halkın büyük bir çoğunluğu Sayın Özal’ı cumhurbaşkanı olarak görmek isteme mekte idiyse, Sayın Özal bunu herkesten iyi bilerek cumhurbaşkanlığına
Bu modellerden hangisinin doğru olduğunu görebilmek için Plüton’un Güneş’ten uzaklaşırken incelenmesinin gerekli olduğunu söyleyen gökbilimciler, NASA’nın New
Cüce Eliptik Gökada Takımyıldız: Andromeda Uzaklık: 2,2 milyon ışık yılı Parlaklık: 8,1 kadir.. M32, Andromeda’nın çok yakınında bulunan küçük ama
Ufkun açık olduğu bir yer- den, Güneş battıktan hemen sonra batı-kuzeybatı ufku üzerinde çok kısa bir süre için görülebilir.. Ay, 3 Temmuz’da yeniay, 10 Temmuz’da
“Nafs al-Amr and the Possibility of Objective Truth: An Introduction to the Problem” adını taşıyan ilk bölüm “Nafs al-Amr and the Meaning of
The phenolic acid is either gallic acid, in the case of gallotannins, or else hexahydroxydiphenic acid (=HHDP) and its oxidized derivatives(dehydrohexahydroxydiphenic acid
Where did you come from?” She took us in, we didn’t speak Turkish, I said to her in Kurdish “They killed everyone, we walked and reached here, and this is my brother.” This