Disponibleenlignesur
www.sciencedirect.com
ORIGINAL
ARTICLE
Effect
of
high
intensity
interval
training
on
elite
athletes’
antioxidant
status
Effet
de
l’entraînement
par
intervalles
à
haute
intensité
sur
le
statut
antioxydant
des
athlètes
de
haut
niveau
A.
Faruk
Ugras
BilkentUniversity,DepartmentofPhysicalEducationandSports,06800Ankara,Turkey Received28July2010;accepted25April2012
Availableonline7June2012
KEYWORDS Athletes; Intervaltraining; SOD; CAT; GPX; MDA Summary
Objective.—Theeffectsofhighintensityintervalexercisesonantioxidantdefensesystemare not clear.Since thereisanevidentlackofstudies focusedonoxidative stressexperienced followingcombatsportsandhighintensityintervaltraining,weinvestigatedoxidativestress markers (malondialdehyde[MDA], catalase [CAT],glutathione peroxidase[GPX], superoxide dismutase[SOD])bycompletinghighintensityintervaltrainingprogram(HIITP)andfollowing InternationalMuayThaiChampionship(IMTC).
Methods.—Thestudywascarriedouton21eliteplayers(15malesandsixfemales)whohad regularexercisingandtraininghabits.Theparticipantsweresubjectedtoadaily3-hourHIITP duringbrieftrainingcamp(10-day)beforeIMTC.Theywereinstructedtomaintaintheirnormal dietarypracticesthroughoutthecampandduringthestudytotakenoantioxidantcontaining vitamintablets.
Results.—TherewasasignificantincreaseinMDAlevelsandsignificantdecreaseinCAT activi-tiesofplayers(P<0.05).ThedifferencesinSODandGPXactivitieswerenotsignificant.
Conclusion.—Theseresultssuggestedthathighintensityintervaltrainingandcompetitioncould affecttheoxidativestatusofMuayThai(MT)athletes.
©2012ElsevierMassonSAS.Allrightsreserved.
MOTSCLÉS Athlètes; Entraînementpar intervalles; SOD; CAT; Résumé
Objectif.—Leseffetsdesexercicesdehauteintensitéparintervallesurlesystèmededéfense antioxydantnesontpasclairs.Commeilexisteunmanqueévidentd’étudesportantsurlestress oxydatifsurvenulorsdessportsdecombatetd’entraînementsparintervallesàhaute inten-sité, nous avonsétudiéles marqueursde stress oxydatif(malondialdehyde[MDA], catalase
[CAT], glutathione peroxidase [GPX], superoxide dismutase [SOD]) en complétant les pro-grammesd’entraînementsparintervalledehauteintensité(EPI)etàlasuiteduChampionnat internationaldeMuayThaï(IMTC).
E-mailaddress:[email protected]
0765-1597/$–seefrontmatter©2012ElsevierMassonSAS.Allrightsreserved. http://dx.doi.org/10.1016/j.scispo.2012.04.009
GPX; MDA
Méthodes.—L’étudeaétéréaliséesur21athlètesdehautniveau(15hommesetsixfemmes) quiavaientl’habituded’effectuerdesexercicesetdesentraînementsréguliers.Les partici-pantsontétésoumisàunprogrammed’entraînementsparintervallejournaliersdetroisheures (EPI)pendantdesstagesd’entraînementdecourtedurée(dixjours)avantleIMTC.Les ath-lètesavaientpourinstructiondemaintenirleurshabitudesalimentairespendanttouteladurée dustaged’entraînementetdeneprendreaucuncomprimédevitaminesantioxydantsdurant l’étude.
Résultats.—Il y aeuuneaugmentation significativedes niveauxde MDAet unediminution significativedesactivitésdeCATdesathlètes(P<0,05).Lesdifférencesdanslesactivitésde SODetdeGPXnesontpassignificatives.
Conclusion.—Cerésultat suggèreque l’entraînementparintervalledehaute intensitéet la compétitionpeutaffecterlestatutoxydatifdesathlètesdeMuayThaï(MT).
©2012ElsevierMassonSAS.Tousdroitsréservés.
1.
Introduction
Reactive oxygen species (ROS) are generated by regular metabolic process in vivo and can initiate a cascade of free-radicalformationanddamagetomacromolecules[1].
Oxidativestressisaninevitableconsequenceofaerobiclife,
andthereisgrowingevidencethattheendogenous
genera-tionofROSplaysamajorroleinagingandmanypathological
conditions[2]. In resting state the bodyis equipped with
both non-enzymatic and enzymatic antioxidant defense
systemtoscavengethepotentially harmfuleffectsofROS
[3,4].Thissystemincludesantioxidantenzymessuchas
glu-tathioneperoxidase(GPX),catalase(CAT),andsuperoxide
dismutase (SOD), and non-enzymatic molecules including
vitaminE,vitaminC,vitaminAprecursor,thiol-containing
compounds e.g. glutathione (GSH). These antioxidant
defense systems preserve homeostasis for normal cell
functionsatrestandundernormalphysiologicalconditions.
However,duringstrenuous exercise, pathogenic processes
and aging, ROS production may overwhelm antioxidant
defense capacity causing cell and tissue damage [5,6].
As consequencesthe bilayerlipid membraneof the cells,
proteins, and even DNA material may suffer oxidative
damage[7].
Manyathletesinvolvedindifferentintensephysical
activ-itiesduringphysicaltrainingandsportiveevents,whichare
performedinuncontrolledsettingandoftenpossess
compo-nentsofbothanaerobicandanaerobicnature[8].Excessive
trainingcanproduce oxidative stressand antioxidant
ele-mentsof organismswereaffected withthischallenge[9].
Fewstudieshaveinvestigatedoxidativestressinresponseto
bothaerobicandanaerobicexercisebouts[10,11]especially
oxidative stress, which is experienced following sporting
competitions [8]. Higher resting plasma MDA levels were
reportedin sprint-trainedathletes comparedwithcontrol
subjects.ItwasreportedthatplasmaMDAlevelsfollowing
anextremeenduranceeventineliteathleteincreased[12].
Severalstudieshavealsoreportedthatacute,highintensity
physicalexerciseinducesoxidativestress[13—16].
Muay Thai (MT) is one of the combat sports and field
observationhasshownthatitisintermittentinnatureand
similartothatofkarate,taekwondo,boxingandwrestling.
MTtrainingconsistsofbothstrenuousandexhaustive
exer-cises[17].MTinvolvesastyleofboxingwherecompetitors
try to win bouts and international level amateur bouts
involve four 2-minute rounds with 1-minute rest periods
[18]. Sparring is very intermittent in nature,divided into
periods of veryhigh intensive activity (whenattacking or
blocking) and low intensive activity when the athlete is
preparing for an attack or just moving around [19] from
which athletes can receive short term recovery, as well
as time to prepare for following attack. Thus, sparring
performances principally rely on the immediate (ATP-PC)
and short-term (Glycolysis) energy systems [17]. As the
developmentoffreeradicalsandscavengingofthemarean
importantconsiderationforoptimalperformance,recovery,
and health for players [20]. We aimed to investigate the
oxidativestatusofMTathletesbycompletinghighintensity
intervaltrainingprogram(HIITP)andfollowingInternational
MuayThaichampionship(IMTC).
2.
Methods
2.1. Subjects
Inthisstudy,15malesandsixfemalesMTnationalathletes
wereselectedastheathleticsubjects.Theoxidativestress
parametersofathletesbeforecampperiodwereusedasa
controlvalueforapropercomparison.Subjectshadregular
exercising and traininghabits for three times a week for
120minutes at an elite level. Each athlete had at least
6 years of trainingexperience. The exclusion criteriafor
studyweredrugsandmedicinesintakeboth,sufferingfrom
someillnessandcigarettesmoke.Noneoftheathleteshad
tobeexcludedfromthestudy.Duringcamps,athleteswent
undersomeof thehematologicaltestsasanormal
proce-dure. The investigatedperiod included the 10 dayscamp
durationbeforetheIMTC.Beforethetests,theparticipants
were given adequate information about the importance
of the study and they signed a consenting document. All
training sessions took place at the same time of day to
controlthecircadianvariationinperformance.Duringthe
study, the subjects stayed in the same hotel and were
providedwiththesamedietwithoutanyextraantioxidants
or other nutritional supplements. The subjects showed
100% compliance withtheexercise training program.The
participants were instructed to refrain from eating or
drinkingimmediatelybeforethetests,andtorefrainfrom
exercise24hoursbeforeeachtestingsession.Theprotocol
startedonedaybeforethebeginningofthetrainingperiod.
2.2. Physiologicalmeasurements
Thebodymasswasmeasuredusingcalibrateddigitalscales
andheightwasmeasuredusingaportablestadiometer.The
age of athletes were accurately recorded in years. Blood
samplesweretakenfromtheparticipantthreetimes; the
firstonepriortotraining,thesecondoneafterthetraining
campandthethirdoneaftertheIMTC.Wealsoanalyzedthe
oxidative stress biomarkers at threedifferent times:
pre-camp,aftercampandattheendoftheIMTC.Thetraining
consistedofexercisestostrengthenphysicalfitnessthrough
technicaltrainingandsparringpractices.TheHIITPapplied
toallathletes. The present HIITPwasdesignedaccording
toeliteMTathletes’needs,aftertakingintoconsideration
‘‘theprerequisitesinperformance’’.TheHIITPisavaluable
methodfor improving both aerobic andanaerobic fitness.
Trainingintensitywasdeterminedbyuseofmaximalheart
ratemethod[21].
The HIIT sessions lasted approximately 3hours a day;
1½hours in the morning, 1½hours in the afternoon for
the 3rd, 5th, 7th, and 9th day of camp. Athletes began
dailypracticesbothinthemorningandafternoonsessions
witha20minuteswarm-up,thatincludedrunning5minutes
andwasfollowed bycalisthenicsandstretchingexercises.
Afterwarm-up,actualpracticeforHIITPapproximatelytook
52 minutes in the morning and 48minutes in the
after-noonincludingavarietyof repeatedkickingand punching
techniques and mini matches for 12minutes only in the
afternoonsessions.Twentyminutescoolingdownexercises
followed after both trainingprograms. For the remaining
days,athletesrunwithaerobicintensityandpracticelight
MTexercises inthe morning.Intheafternoonallathletes
takeacompleterest.Duringtrainingcamp,alongwiththe
pre-competitiontrainingroutine,mentalandpsychological
skills(emotionalcontrol,arousalmanagementi.e.)tailored
towardsthespecificneedsofathletesandtheypracticedat
thesimulatedcompetition.
2.3. Trainingsession—Highintensityinterval trainingprogram(HIITP)
2.3.1. Morningpractices
Technicalworkout(TW):(frontkick,sidekick,roundhouse
kickandpunches-alldonewithalternatinglegsandarms):
• 20minuteswarm-up;
• 6secondswork(95—100%ofMaxVO2),18secondsrest,20
rpt,totaloftwosetsofwork,5minutesrest1inbetween
theset;
• 10secondswork(95—100%ofMaxVO2),30secondsrest,12
rpt,totaloftwosetsofwork,5minutesrest1inbetween
theset;
• 20minutescoolingdownexercises.
1Passiverecoveryreferstoperiodsthatdonotinvolveanyform
ofactivity.
2.3.2. Afternoonpractices
TW:
• 20minuteswarm-up;
• 30secondswork(80—90%ofMaxVO2),60secondsrest,4
rpt,totaloftwosetsofwork,3minutesrest2inbetween
theset;
• 60secondswork(80—90%ofMaxVO2),120secondsrest,2
rpt,totaloftwosetsofwork,3minutesrest2inbetween
theset;
• 2minutes sparring, total of foursets of work, 1minute
restinbetweentheset;
• 20minutescoolingdownexercises.
2.3.3. Trainingprotocol
Period:10-day.
Trainingfrequency:2timesaday.
1stday:1sttest.
2nd,4th,6th,8thdays:½dayaerobicrunningandlow
intensityMTtechnical/tacticalactivities&½dayrest,3rd,
5th,7th,9thdays:HIITP.
10thday:2ndtest.
Traininghours:10.00—11.30a.m.and5.00—6.30p.m.
Trainingtype:Interval(everyotherday)andaerobiclow
intensityofrunning.
Training intensity: 95—100% of MaxVO2 (Morning) and
80—90%ofMaxVO2(Afternoon).
Trainingperiod:approximately1½hour/session.
BloodTest: 1st,10th days of the camp, and after the
championship.
2.4. Measurementofoxidativestatus
Erythrocyte SOD, GPX, and malondialdehyde (MDA) were
measured as previously described by Aydin et al. [23].
Erythrocyte CAT activity was measured in hemolysates as
describedbyAebi[24].
2.5. Statisticalanalysis
Allstatisticalanalyses werecalculatedbythe SPSS
statis-ticalpackage. A paired t-test wasused todetermine the
differencesin physiological parameters between pre-and
post-values.Datawereexpressedasmeanvalues±standard
deviation(SD)andstandarderror(SE).Differencesbetween
beforeandafter exercises werereported asmean
differ-ence±95%confidenceintervals.Thestatisticalanalysiswas
calculatedwithnon-parametrictestWilcoxonSignedRanks
testfortheoxidativestressparameters.
3.
Results
PhysicalcharacteristicsoftheMTmaleandfemaleathletes
are presented in Table 1. The body weight of male and
femaleathletesdidnotchangedduringthechampionship.
2Active rest-lowto moderateactivityis effectiveimmediately
followingsustainedhighintensitytrainingorcompetition, partic-ularly where the anaerobic glycolytic energy pathway has been substantiallyinvolved[22].
Table1 PhysicalcharacteristicsoftheMuayThai(MT)maleandfemaleathletes.
SelectedPhysicalParameters BeforeX1 AfterX2 Meandifference±95%CI
Male(n=15) Age(year) 20.93±2.43 Height(cm) 178.60±6.98(1.80) Bodyweight(kg) 68.71±13.02(3.36) 68.04±12.72(3.28)* 0.67±0.50(0.13) Female(n=6) Age(year) 18.83±1.47 Height(cm) 170±1.79(0.73) Bodyweight(kg) 57.28±7.58 56.86±7.59(3.09)* 0.41±0.09(0.04)
Valuesarethemean±SD(SE).Beforethecamptraining;afterthecamptraining*(P<0.01).
Table2 TheerythrocyteoxidativestressstatusofMuayThai(MT)athletes.
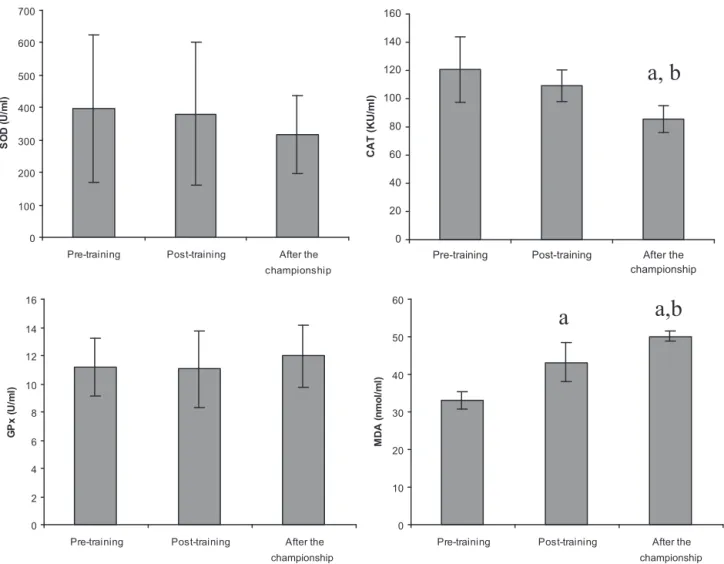
SOD(U/ml) CAT(KU/ml) GPX(U/ml) MDA(nmol/ml) Pre-training 343.62±168.38 111.06±22.28 11.45±3.12 32.67±2.47 Post-training 330.10±168.40 109.30±17.12 10.76±3.18 45.52±4.01a Afterthechampionship 293.65±128.37 87.07±13.69a,b 11.61±3.07 49.80±2.29a,b SOD:superoxidedismutase;CAT:catalase;GPX:glutathioneperoxidase;MDA:malondialdehyde.n=21(sixfemales,15males).
ap<0.05whencomparedwithpre-training. b p<0.05whencomparedwithpost-training.
0 50 100 150 200 250 300 350 400 450 500
After the championship Post-training Pre-training SO D (U /m l) 0 20 40 60 80 100 120 140 After the Post-training Pre-training championship CA T ( K U/ m l) 0 2 4 6 8 10 12 14 16 After the Post-training Pre-training championship G P x ( U /m l) 0 10 20 30 40 50 60 After the Post-training Pre-training championship M DA (n m ol /m l)
a, b
a, b
a
Figure1 Theoxidativestressstatusofmaleathletes.Threedependentgroupswerecomparedwithnon-parametricWilcoxon SignedRankstest.Valuesareexpressedasmean±S.D.SOD:superoxidedismutase;CAT:catalase;GPX:glutathioneperoxidase; MDA:malondialdehyde.n=15.a:P<0.05whencomparedwithpre-training;b:P<0.05whencomparedwithpost-training.
0 100 200 300 400 500 600 700
Pre-training Post-training After the championship
S O D (U /m l) 0 20 40 60 80 100 120 140 160
Pre-training Post-training After the championship CA T (KU /m l) 0 2 4 6 8 10 12 14 16
Pre-training Post-training After the championship GP x (U /m l) 0 10 20 30 40 50 60
Pre-training Post-training After the championship MD A (n m ol /m l)
a, b
a,b
a
Figure2 Theoxidativestressstatusoffemaleathletes.Threedependentgroupswerecomparedwithnon-parametricWilcoxon SignedRankstest.Valuesareexpressedasmean±S.D.SOD:superoxidedismutase;CAT: catalase;GPX:glutathioneperoxidase; MDA:malondialdehyde.n=6.a:P<0.05whencomparedwithpre-training;b:P<0.05whencomparedwithpost-training.
OxidativestressparameterswerepresentedinTable2.MDA
levelsincreased post trainingand after thechampionship
(P<0.05),CATactivitiesdecreasedafterthechampionship
(P<0.05). SOD activities decreased post training and
after the championship, but these differences were not
significant. GPX activities did not change. The oxidative
stress status changed for both female and maleathletes
(Table2,Figs.1and2).
4.
Discussion
Physicalexerciseisacomplexbiologicalactivitychallenging
homeostasisatthecell,tissue,organandwholebodylevels
[25].There arenumerousreports that providereasonable
supporttothenotionthatexerciseincreasestheproduction
of ROS [26]. Little is known, regarding the extent of
oxidative stress when comparing aerobic and anaerobic
exercisemodes[10].
On theoccurrenceofexercise,stress isnotfullyclear.
However, the principal factor responsible for oxidative
damage during exercise is the increase in oxygen
con-sumption[27],Itappearsthatanaerobictypesofexercise,
whichinvolveslessoxygencirculationthroughoutthebody
thanaerobicexerciseis associatedwithan increasedROS
generationlevel throughotherpathways [8,11]suggesting
thatoxygenconsumptionper seisnotthemajorcauseof
exercise-inducedoxidativedamage[11].
Differenttypesofexercisewouldhavedifferenteffects
onoxidative stress [11] which is definedas asituation in
whichanincreasedlevelofROSgenerationoverwhelmsthe
antioxidantdefensecapacity,resultinginoxidativedamage
tolipids,proteinsandDNA[11,28].
A review of literature on changes in oxidative stress
markersandphysicalparametersfollowingtheMTtraining
and championship indicates a lack of information in this
subjectarea.Forthisreasonweinvestigatedtheoxidative
statusofathletesduringcompetitionseasonbycompleting
HIITprogramandfollowingIMTC.
Field observation has shown that, MT activity is
extremely dynamic in nature and during sparring athlete
repeatedexplosivemovementsatahighintensityfollowed
by sub-maximal work. Thus the primarily energy systems
utilizedaretheanaerobicATP-CPandlacticsystems[17].
Itwasreportedthattheactivitiesofantioxidantenzymes
anacuteboutofexerciseinskeletalmuscle,heartandliver
[3].
SOD,CAT,GPXanMDAactivitiesinresponsetoexercise
arevariable. When we looked at our study, we observed
thatMDAlevelsincreasedintheperiodofposttrainingand
afterthechampionship(P<0.05),CATactivitiesdecreased
afterthechampionship(P<0.05).SODactivitiesdecreased
in the post-training period and after the championship,
but these differences were not significant. GPX activities
didnotchange inall samples.The oxidative stress status
changedforbothfemaleandmaleathletes.Itisconcluded
withtheseresultsthatoxygenradicalswereproducedafter
strenuous physical training and some antioxidant enzyme
activitiesweredecreased.
When oxygen radicals were not scavenged properlyby
antioxidantenzymestheyaffected lipidmolecules incells
andMDAlevelswereincreased.Theseresultssupportedour
hypothesis.Zoppietal.[7]arrivedatthesameconclusion
intheirstudy,suggestingthatantioxidantsupplementation
hadpositiveeffectsonantioxidantcapacityoftheplayers.
Baker et al. [29] reported the increased MDA levels in
high-intensityexercise and supported ourresults. Several
studiesreportedthatsingleboutofexerciseincreaseblood
levelsofMDA [4,14].Higher resting plasmaMDA reported
insprint-trainedathletes comparedwithcontrolsubjects.
Elevated in plasma MDA following an extreme endurance
event in elite athlete also reported [12]. Not all studies
reportedincreases in MDA in response to physical effort.
MiyazakiH.etal. observed nochange in erythrocyte MDA
aftera12-weektrainingprogram[14].
According to the Metin et al. [9] excessive training
can produce oxidative stress and organism’s antioxidant
elements were stimulated with this challenge. This idea
canbeexplainedasanadaptivemechanism.Inevery
condi-tion it is clear that oxygen radicals are produced in the
organismsduringstrenuoustrainingperiods.Forthisreason
nutritionplanningincludingantioxidantsupplementationis
consideredasanimportantmeasureagainstthehazardous
effectsoffreeradicals.
SODisoneofthemainantioxidantenzymesthatdegrade
superoxide radicals [30] increase in SOD enzyme activity
correspondswith enhancedresistance tooxidative stress.
Groussardet al. found that SOD activity decreased after
a single sprint anaerobic exercise [27]. Not all studies
reported decrease in SOD in response to exercise. It has
been reportedthat 8-week moderate intensity of aerobic
trainingdid notelevate SOD activity.Furthermore,it has
beenrevealedadecreaseinSODlevelsfollowingan acute
boutofexerciseinskiersparticipatinginagradedtreadmill
test to exhaustion and elevated erythrocyte SOD activity
immediatelypost-exercisewhenthesprintersperformeda
sprintexercise[12].
CATactivityinresponsetoexerciseisvariable.Following
about of sub-maximalexercise adecreasein erythrocyte
CATactivityreportedintrainedcyclists.
Furthermore, it has been reported that sprinters who
performedasprint-typeexercisedidnothavealtered
ery-throcyteCATactivity[12].
GPXactivityisakeycomponentoftheglutathione
home-ostasisanditsresponsetoexerciseisvariable[28].Higher
inoxygenconsumptionduringexerciseactivatestheenzyme
GPXtoremovehydrogenperoxide.Inresponsetoanacute
bout of high intensity exercise, elevated erythrocyte GPX
activityhasbeenfoundafterasprintexercisebutnochange
whenrunnersperformedanenduranceexercise[12].
The present study is the first to report improvements
in oxidativestatus aftershort-termhighintensity interval
trainingandimmediatelyafterMTcompetition.We
specu-latethat changesintheseparametersmight representan
increaseinROSafterhighintensityintervaltraining.
Incomparisonwithother investigators, webelieve the
presentstudyprovidedthefirstdirectanalysisofeffectof
highintensityintervaltrainingoneliteathletes’antioxidant
statusaftertrainingandcompetition.
Theresultsofthisstudyalsosuggest thatabrief
train-ingcamp,whichtheHIITprogramis carriedoutappearto
causepositivechangesonphysiological parametersdueto
somebiologicalreasonslinkedwithpreparationforhigh
per-formanceandoxidativestatusof MTathletes.However,it
wasobservedthatoxidativedamageincreased.The
recruit-ment of alimited number ofspecial MT athletes wasthe
limitationofourstudy.Furtherstudiesareneededwhether
similaradaptationsaremanifestafteracoupleofweeksof
differenttypeofintervaltraining.Afterthiskindofstudies
thenutritioncomponentofeliteathletescanbearranged
againstoxidativedamage.
Disclosure
of
interest
Theauthordeclaresthathehasnoconflictsofinterest
con-cerningthisarticle.
Acknowledgements
TheauthorwouldliketothankHuseyinG.Sonmez3for
tech-nical assistance, Ayse Eken, Ph.D.4, Onur Erdem, Ph.D.4,
Cemal Akay, Ph.D.4, Ahmet Sayal, Ph.D.4, Ahmet Aydin,
Ph.D.5, and Mesut Akyol, Ph.D.6 for measuring oxidative
stressparameters,andJuliaGoggin7forhelpingwithproof
reading.
References
[1]Forsberg L, de Faire U, Morgenstern R. Oxidative stress, humangeneticvariation,anddisease.ArchBiochemBiophys 2001;389:84—93.
[2]Cooke M, Evans M, Dizdaroglu M, Lunec J. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J 2003;17:1195—214.
[3]Banarje AK, Mandal A, Chanda D, Chakraborti S. Oxi-dant, antioxidant and physical exercise. Mol Cell Biochem 2003;253:307—12.
3BilkentUniversity,DepartmentofPhysicalEducationandSport,
06800Ankara,Turkey.
4GülhaneMilitary MedicalAcademy,DepartmentofToxicology,
Etlik,06010Ankara,Turkey.
5YeditepeUniversity,FacultyofPharmacy,Departmentof
Toxi-cology,Kayisdagi,34755Istanbul,Turkey.
6GülhaneMilitaryMedicalAcademy,DepartmentofBiostatistics,
Etlik,06010Ankara,Turkey.
7Bilkent University, Faculty Academic English Program, 06800
[4]KoskaJ,Blazicek P,MarkoM,GrnaJD, KvetnanskyR, Oigas M.Insulin,catecholamines,glucoseandantioxidantenzymes inoxidativedamageduringdifferentloadsinhealthyhumans. PhysiolRes2000;49(Suppl.19):S95—100.
[5]JiLL. Free radicalsand exercise:implication inhealth and fitness.JExercSciFitness2003;1(1):15—22.
[6]KanterM.Freeradicals,exerciseandantioxidant supplemen-tation.ProcNutrSoc1988;57:9—13.
[7]ZoppiCC,HohlR,SilvaFC,LazarimFL,NetoJMFA,Stancanneli M,etal.VitaminCandEsupplementationeffectsin profes-sionalsoccerplayers underregulartraining.JIntSocSports Nutr2006;3(2):37—44.
[8]Fisher-WellmanK,BloomerRJ.Acuteexerciseandoxidative stress:a30-yearhistory.DynMed2009;8(1):1—25.
[9]MetinG,GumustasMK,UsluE,BelceA,KayseriliogluA.Effects ofregulartrainingonplasmathiols,malondialdehydeand car-nitineconcentrations inyoungsoccerplayers.ChinJPhysiol 2003;46(1):35—9.
[10] BloomerRJ,SmithWA.Oxidativestressinresponsetoaerobic andanaerobicpowertesting:Influenceofexercisetrainingand carnitinesupplementation.ResSportsMed2009;17:1—16. [11] ShiM, Wang X, Yamanaka T, Ogita F,Nakatani K, Takeuchi
T. Effects of anaerobic exercise and aerobic exercise on biomarkers of oxidative stress. Environ Health Prev Med 2007;12:202—8.
[12] UrsoML,ClarksonPM.Oxidativestress,exerciseand antioxi-dantsupplementation.Toxicology2003;189:41—5.
[13] BelviranlıM,GokbelH.Acuteexerciseinducedoxidativestress andantioxidantchanges.EurJGenMed2006;3(3):126—31. [14] MiyazakiH,Oh-ishiS,OokawaraT,KizakiT,ToshinaiK,HaS,
etal.Strenuousendurancetraininginhumansreduces oxida-tivestress following exhaustingexercise. EurJApplPhysiol 2001;84:1—6.
[15] PepeH,BalciSS,RevanS,AkalinPP,KurtogluF.Comparison ofoxidativestressandantioxidantcapacitybeforeandafter runningexercisesinbothsexes.GendMed2009;6(4):587—95. [16] Sureda A, Tauler P, Aguilo A, Cases N, FuentespinaE, Cor-dovaA,etal.Relationbetweenoxidativestressmarkersand antioxidantendogenousdefensesduringexhaustiveexercise. FreeRadicRes2005;39(12):1317—24.
[17] McArdleWD,KatchFI,KatchVL.Nutritionforphysicalactivity. Exercise training and functional capacity. In: McArdle WD,
KatchFI,KatchVL,editors.Essentialsofexercisephysiology. 1stedPhiladelphia:LeaandFebiger;1994.
[18]MyersTD,BalmerNJ,NevillAM,NakeebY.Evidenceof nation-alisticbiasinMuayThai.JSportsSciMedCSSI2006:21—7. [19]NunanD.Developmentofasportsspecificaerobiccapacitytest
forkarate—apilotstudy.JSportsSciMedCSSI2006:47—53. [20]VollaardNB,ShearmanJP,CooperCE.Exercise-induced
oxida-tivestress:myths,realitiesandphysiologicalrelevance.Sports Med2005;35(12):1045—62.
[21]FoxEL,BowersRW,FossML.Methodsofphysicaltraining.In: FoxEL,BowersRW,FossML,editors.Thephysiologicalbasis for exercise and sport.4thed.New York:SoundersCollege Publishing;1988.
[22]ClementD,DavidsonR,MathesonGO,NewhouseI,SawchukL, CoxD,etal.Issuesspecifictowomen.In:JacksonR,FitchK, O’BrienM,editors.Sportsmedicinemanual.1sted.Canadian CataloguinginPublishingData:Calgary;1990.
[23]AydinA,HilmiO,SayalA,OzataM,SahinG,IsimerA. Oxida-tive stress and nitric oxide related parameters in type II diabetesmellitus:effects ofglycemiccontrol.ClinBiochem 2001;34:65—70.
[24]AebiH.Catalaseinvitro.MethodsEnzymol1984;105:121—6. [25]Ji LL, Radak Z, Goto S. Hormesis and exercise: how the
cell copes with oxidative stress. Am J Pharmacol Toxicol 2008;3(1):44—58.
[26]PackerL,CadenasE,DaviesKJA.Freeradicalsandexercise: anintroduction.FreeRadicBiolMed2008;44:123—5. [27]GroussardC,Rannou-BekonoF,MacheferG,ChevanneM,
Vin-centS,SergentO,etal.Changesinbloodlipidperoxidation markersandantioxidantsafterasinglesprintanaerobic exer-cise.EurJApplPhysiol2003;89:14—20.
[28]KinnunenS, AtalayM,HyypaS, LehmuskeroA,Hanninen O, Oksala N. Effects of prolonged exercise on oxidative stress andantioxidantdefenseinendurancehorse.JSportsSciMed 2000;4:415—21.
[29]Baker JS,Bailey DM, Hullin D,Young I,Davies B.Metabolic implications of resistiveforce selection for oxidative stress and markers ofmuscle damage during30s of high-intensity exercise.EurJApplPhysiol2004;92(3):321—7.
[30]QiaoD,HouL,LiuX.Influenceofintermittentanaerobic exer-ciseonMousephysicalenduranceandantioxidantcomponents. BrJSportsMed2006;40:214—8.