The Donnan Potentials of Polysulfonated Cation Exchange Membranes
Tam metin
Şekil
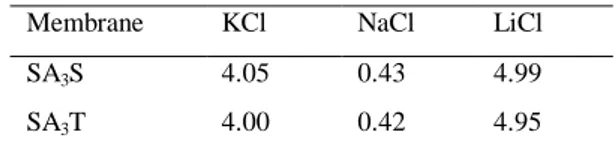
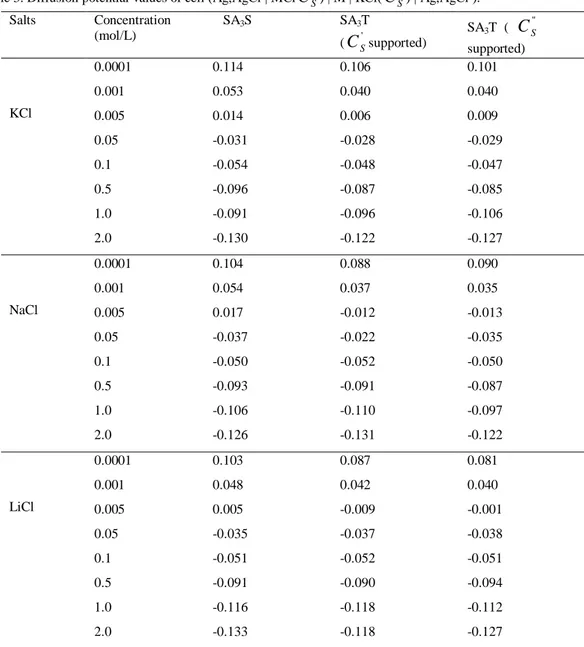
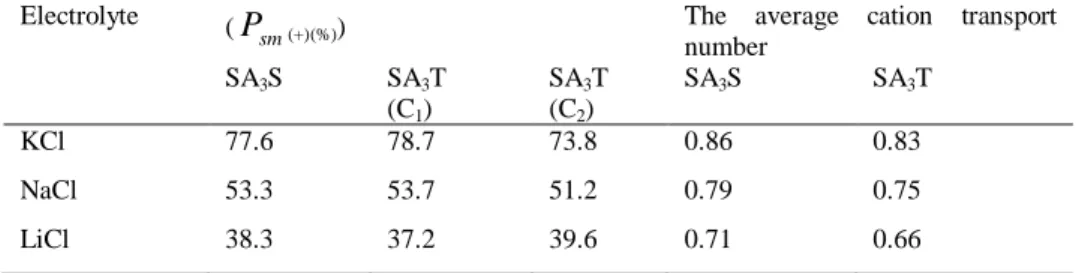

Benzer Belgeler
In situating Dickinson alongside other historical nonconformists, Howe’s study resonates with Hartman’s utopian history of the “chorus.” Howe counters normative narratives that
Heart rate of patients (Beat/min) (Mean+SD). ALFENTANIL DILTIAZEM
Yapılan deneylerde 6 mm kalınlığa, 80 cm ve 40 cm uzunluklara sahip demir levhalar üst üste konularak yapay çatlak modelleri oluşturulmuştur. Doğada değişik şekillere sahip
Tablo 1’de yer alan analiz sonuçlarına göre araştırmaya katılan çalışanların duygusal tükenmişlik ile duyarsızlaşma düzeylerinin düşük düzeyde olduğu, kişisel
Although 3,5-DMAP caused a dose-dependent increase in the mutation frequency, particularly in both TA98 (without S9 mix) and TA100 strains (without S9 mix), the increases were
Kuvveler arasındaki uzlaşım ve uyumun sağlanması ruhsal tıbbın ve ahlaki dengenin kazanılması olduğu için İslam Felsefesinde nefsin kuvve- leri konusu ahlak
Özellikle oksidasyon göstergesi olan TBA açısından ektraksiyon grupları ile kontrol grubu ara- sında önemli bir fark bulunurken, mikrobiyolojik açıdan önemli