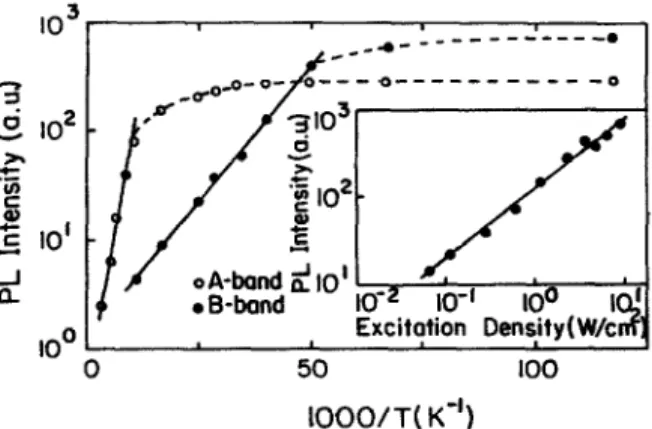
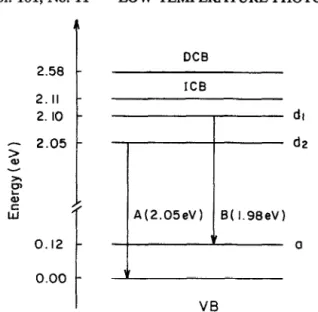
Low temperature photoluminescence spectra of InS single crystals
Tam metin
Şekil
Benzer Belgeler
As a conclusion, in this study the core protein of the hepatitis C virus was cloned, expressed and purified in order to test the human sera with this viral
The gene expression levels of smurf2 and its interacting partners were analyzed with qRT-PCR to reveal possible age- and region-specific patterns and the data showed that while
Abstract We previously showed that the expression of transient receptor potential canonical (TRPC)6 ion channel elevated when TRPC1 was knocked down in A7r5 cultured vascular
Resurgent nationalism is a conspicuous phenomenon in contemporary China as the rhetorical fervor and social pervasiveness of the nationalist discourse among the people,
Chemical information (i.e. distinct Raman bands) are observed to be present in the SERS signal (as observed in com- plementary confocal Raman measurements recorded on the samples
However, for improved energy efficiency of VLP systems, the total power consumption of LEDs must also be taken into account in addition to localization performance requirements
Bu şekilde romanın sonunda büyük bir amaç peşinde olan erkek karakter, Galip, amacına ulaşırken; kadın karakter Rüya, Pamuk’un diğer romanlarındaki kadınlar
This was the main motivation for Carlson to study the productive elements since, when there is a product structure on H ∗ (D ∞ ζ ), it is easier to calculate the differentials in