Different properties of chicken and turkey breast fillets marinated with fruit
juices
Article in Fleischwirtschaft -Frankfurt- · January 2020
CITATIONS 0
READS 132
4 authors:
Some of the authors of this publication are also working on these related projects:
LecturerView project
Technological and textural properties of spent chicken breast, drumstick and thigh meats as affected by marinating with pineapple fruit juiceView project Kubra Unal Selcuk University 12PUBLICATIONS 31CITATIONS SEE PROFILE Halime Alp Selcuk University 2PUBLICATIONS 7CITATIONS SEE PROFILE
Ali Samet Babaoğlu
Selcuk University 9PUBLICATIONS 25CITATIONS SEE PROFILE Mustafa Karakaya Selcuk University 47PUBLICATIONS 407CITATIONS SEE PROFILE
All content following this page was uploaded by Ali Samet Babaoğlu on 02 July 2020.
This research determined the effects of three different dark coloured marinades containing black mulberry (MJ), grape (GJ) and pomegranate juices (PJ) on pH value, water holding capacity (WHC), cooking loss (CL) and colour (L*, a*, b*) properties of chicken and turkey breast meats. Lipid oxidation (Thiobarbituric acid value-TBA), antioxidant activitiy (DPPH-2,2-diphenyl-1-picrylhydrazyl), textural properties (Meullenet-Owens razor shear force and energy) and sensory analyses of the samples were also performed. Fruit juices generally decreased the pH and WHC values compared to control. The meat samples treated with MJ had the highest a* value and the marination with PJ increased the b* values of samples. Turkey breast samples marinated with GJ were determined to be the most tender. Marinating with fruit juices significantly inhibited lipid oxidation and increased (p < 0.01) the DPPH values in chicken and turkey meats. These natural marinades should be recommended to develop consumer acceptability of chicken and turkey breast meat samples.
P
oultry meat is very important for human nutrition because of provid-ing high-quality protein and low levels of fat, with a desirable fatty acid profile (ANANGet al., 2010; RADHAKRISHNANet al., 2014). Productionand consumption of poultry meat have increased rapidly in the last years (FAO, 2018). Demand for poultry meat is expected to continue increasing due to population growth, low production costs and more affordable animal protein compared to red meats (FAO, 2017). Also, the demand for marinated chicken products has been growing continu-ously in many parts of the World (WHITEand KOKOTSAKI, 2004).
In meat industry, marination has been applied to improve flavor, tenderness and increase yield of raw meat (ALVARADOand MCKEE, 2007).
Additionally, due to the presence of antioxidative and antimicrobial substances in the formulation, marination can also prolong the shelf life of meat, delaying lipid oxidation and growth of spoilage micro-organisms (KARGIOTOUet al., 2011; NISIOTOUet al., 2013; POKORNY, 1991).
Traditionally a marination contains ingredients, such as lemon, wine, vinegar, organic acids, essentials oils, spices, herbs and salt (LYTOUet
al., 2017; PATHANIAet al., 2010). Nowadays, fruit juices such as
pome-granate, pineapple or kiwi and also alcoholic beverages like whiskey, brandy can also be used for marination of meat (LYTOUet al., 2017). After
the meat is dipped, marinade components passively diffuse the meat during storage (ALVARADOand MCKEE, 2007).
So far, the most commonly used poultry marinades has included water, salt, sodium tripolyphosphate, sodium lactate, sodium citrate, lemon, and vinegar (ALVARADOand MCKEE, 2007; Yusop, 2011). However,
limited number of studies in poultry meat have been reported about marination with fruit juices such as black grape, black mulberry and pomegranate juice (GÖKand BOR, 2016; VAITHIYANATHANet al., 2011).
Black grape juice has antioxidant properties due to presence of phenolic compounds (FULEKIand RICARDO-DA-SILVA, 2003; PARKet al.,
2003). Mulberry (Morus alba) is one of the most important fruits have large amounts of phenolic compounds, total flavonoid, ascorbic acid, mineral and amino acid (HANet al., 2017; LEEand HWANG, 2017).
Antho-cyanins contribute the purple-black colour and constitute the major flavonoid group in black mulberry (FAZAELIet al., 2013). Therefore, black
mulberry juice could be used as an alternative natural antioxidant to improve quality of meat product (CHENGet al., 2016; SLAVINet al., 2013).
Pomegranate (Punica granatum L.), is a fruit, has contained bioactive compounds. Therefore, pomegranate juice has been demonstrated to be high in antioxidant activity (ADHAMIand MUKHTAR, 2006). Hence the
purpose of this study was to investigate the effects of marination with black grape, black mulberry and pomegranate juice on the physico-chemical, textural and sensory properties of chicken and turkey breasts.
Materials and methods
Sample preparation
Chicken and turkey meats were provided from a local farmer in Konya, Turkey. The carcasses were transported to the laboratory within a maximum of 25 min and then shredded, according to standards. The breast of each chicken and turkey were manually separated from car-casses and were packed separately. Each breast pieces were stored in refrigerated units at 4 °C for 18 h.
The fresh fruits of black mulberry (Morus nigra), grape (Vitis vinifera) and pomegranate (Punica granatum ) to be used for marination were procured from a local market. On the day of the analysis, the each fruits were manually peeled and pressed to the fruit juice.
Marination
Existing skin and excess fatty parts were removed from the chicken and turkey pieces before marination. Containers with a depth of 10 cm and a width of 40 cm were filled with fruit juice, for marination pro-cessing. The each meat samples were kept in the marination solution for 72 hours at 4 °C.
pH values
The pH values of the samples were measured with a pH meter (WTW 315 i set model, Weilheim, Germany) following the instructions as outlined (AOAC, 2000).
Colour properties
Colour measurements were performed using a CR-400 chromameter (Konica Minolta, Inc., Osaka, Japan) with illuminant D65, 2° observer
Different properties of chicken and turkey
breast fillets marinated with fruit juices
Keywords
»
Breast meat
»
Fruit juices
»
Marination
»
Texture
Received: 29 May 2019 | reviewed: 7 June 2019 | revised: 4 October 2019 | accepted: 11 January 2020
Forschung und Entwicklung
angle, diffuse/O mode, 8 mm aperture for illumination and 8 mm formeasurement. The instrument was pre-calibrated against a white reference tile (L* = 97.10, a* = 4.88, b* = 7.04). The American Meat Science Association (AMSA) guidelines for colour measurements were followed (HUNT et al., 1991).
Cooking loss (CL)
The CL of marinated meat samples were measured as outlined by KONDAIAH et al. (1985). Breast meat samples of 20 g were placed in a
polyethylene bag and heated in a water bath at 80 °C to achieve an internal temperature of 72 °C.
Water holding capacity
Tubes containing an aliquot of the marinated meat sample (8 g) and 12 ml of 0.6 M NaCl solution were placed into a 5 °C water bath for 15 min and then centrifuged (4 °C) at 10000 rpm for 15 min. The super-natant was recovered, to determine the water holding capacity (WHC, %) of the meat batches (WARDLAW et al., 1973).
Texture measurement
For the cooking process, chicken and turkey meat samples were cooked in a convection oven at 150 °C for 30 min until an internal tem-perature of 76 °C was reached (SAHA et al., 2009). The temperature was
measured using a thermometer (Digitale Bratengabel-TCM). Once cooked, the samples were cooled down to about 25 °C before analysis.
The textural properties of the samples were determined using a TA-HD Plus texture analyser (UK) equipped with a Meullenet-Owens razor shear and a 50 kg load cell. A certain number of readings were made from specific regions of the samples using the software program of the texture measuring device (CAVITT et al., 2004).
TBA values
The method described by OCKERMAN (1985) was used to determine the
extent of oxidative rancidity of the marinated poultry samples. The absorbance was read at 538 nm (UV-160 A, UV-Visible Recording Spec-trophotometer, Shimadzu, Tokyo, Japan) against a reagent blank. The TBA numbers were expressed as milligrams malonaldehyde per kilogram samples.
Antioxidant activity
measurement
Antioxidant capacity was deter-mined using a modified method of BRAND-WILLIAMSet al. (1995). The
absorbance of the solution was monitored at 517 nm. The standard curve was developed with ascor-bic acid and DPPH.
Sensory evaluation
The meat samples were evaluated by nine experienced panellists, for meat apperance, texture, flavour and overall acceptance, using a 9-point scale (1 = “the clearest colour, the toughest, the lowest flavor and the lowest overall acceptability; 9 = “the darkest colour, the most tenderness and the highest overall acceptability”).
Statistical analysis
To evaluate the effect of the marination time on the parameters studied, the collected data were examined by analysis of variance (ANOVA), using MINITAB for
Win-dows Release 16.0. Each parameter was tested in triplicate samples with two replications.
Results and discussion
pH, cooking loss and water holding capacity
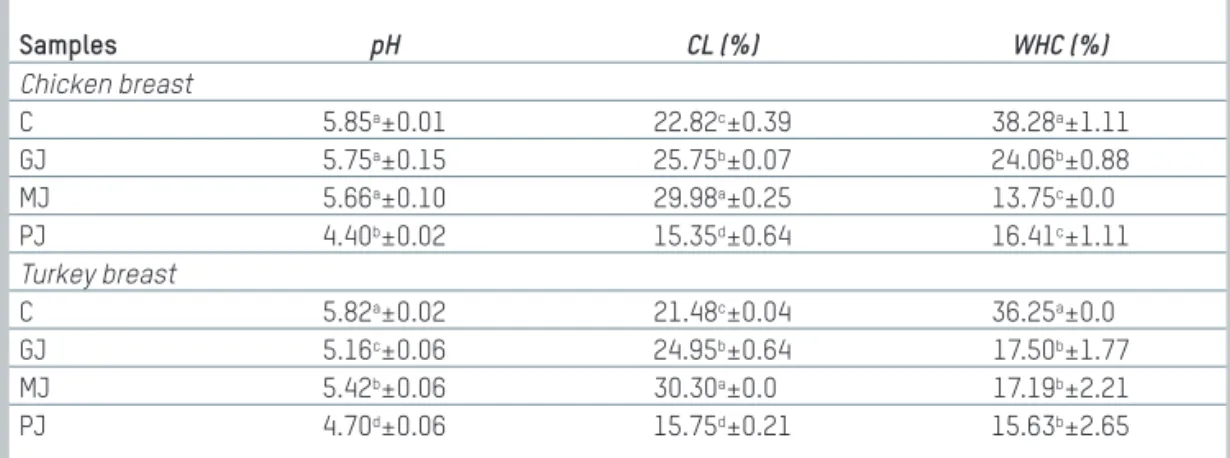
Table 1 shows pH, cooking loss (%) and water holding capacity (%) values of chicken and turkey meat samples treated with distilled water (control sample; C), grape (GJ), black mulberry (MJ) and pomegranate juices (PJ).
GJ, MJ, PJ have a significant effect (p < 0.01) on the pH values of chicken and turkey meat samples. Before the chicken and turkey breast meats marinated with fruit juice, the pH values were about 5.89 and 5.81. The pH values of marinades were 3.86, 3.96 and 3.19 for GJ, MJ, PJ, respectively. Marinating meat samples with fruit juices generally decreased the pH values compared to controls and the reduction varied among the various treatments. SERDAROĞLUet al. (2007) recorded that
the acidic marinades reduced meat pH. As expected, the low pH of PJ significantly reduced the pH of marinated chicken and turkey meat samples compared to the other treatments (Tab. 1). The pH values of chicken and turkey meat samples treated with PJ were determined as 4.40 and 4.70. Similarly, FIRUZIet al. (2019) stated that the pH values of
the frankfurter samples containing PJ concentrates were lower com-pared to the control sample at the beginning of the storage process. The authors claimed that this could result from the acidic pH of pome-granate juice concentrates.
DEVATKALet al. (2010) informed that the pH values of cooked goat
patties added kinnow rind, pomegranate rind and powder extract were significantly lower than the control sample. This result may be based on the acidic pH of fruit extracts added to patties.
On the other hand, KURT(2017) recorded that the effects of
pomegran-ate seed flour on the pH value of raw beef patties was not significant. Similarly, NAVEENAet al. (2008) reported that the addition of pomegranate
juice extract to chicken patties presented no significant difference in the pH values of cooked patties. VAITHIYANATHANet al. (2011) observed that
the pH values of chicken breast samples dipped in pomegranate fruit juice phenolic solution slightly increased from 5.87 to 5.91 in the middle of the storage days. The authors reported that the treatment meat samples with pomegranate phenolic solution resulted in increases
....
....
...
...
...
...
...
...
....
....
....
....
....
....
....
Tab. 1: Average pH, cooking loss and water holding capacity of chicken and turkey
breast samples
Tab. 1: Durchschnittlicher pH-Wert, Kochverlust und Wasserhaltevermögen von Hühner- und Putenbrustproben Samples pH CL (%) WHC (%) Chicken breast C 5.85a±0.01 22.82c±0.39 38.28a±1.11 GJ 5.75a±0.15 25.75b±0.07 24.06b±0.88 MJ 5.66a±0.10 29.98a±0.25 13.75c±0.0 PJ 4.40b±0.02 15.35d±0.64 16.41c±1.11 Turkey breast C 5.82a±0.02 21.48c±0.04 36.25a±0.0 GJ 5.16c±0.06 24.95b±0.64 17.50b±1.77 MJ 5.42b±0.06 30.30a±0.0 17.19b±2.21 PJ 4.70d±0.06 15.75d±0.21 15.63b±2.65
Values correspond to mean data ± corresponds to standard deviation.a–dWith different letters in columns are significantly different (p < 0.01). C: Control (Distilled water), GJ: Marination with black grape juice, MJ: Marination with black mulberry juice,
PJ: Marination with pomegranate juice
Source: ÜNALet al. FLEISCHWIRTSCHAFT 2_2020
(p < 0.05) of pH values with storage period and the storage of meat samples at 0 – 4 °C temperature causes lots of chemical reactions.
Table 1 indicates cooking loss (CL, %) and water holding capacity (WHC, %) values of chicken and turkey meat samples marinated with distilled water (control sample), grape, black mulberry and pomegran-ate juices. WHC is one of the important quality criteria in meat technol-ogy and determines the quality characteristics of meat and meat products such as colour, structure and tenderness. The chicken sam-ples treated with fruit juice had lower water holding capacity (WHC) values than the control samples because the pH values of treated samples were lower than the control samples (Tab. 1). Additionally, there is a significant difference (p < 0.01) between the control, grape juice treated and the other treated chicken samples, and MJ and PJ
treated samples had the lowest WHC values (13.75%, 16.41%). The cooking loss values of chicken and turkey meat samples marinated in PJ were the lowest as compared to the other samples. In contrast, marination in GJ and MJ increased the CL values of meats than the control. KURT(2017) recorded that pomegranate seed flour didn’t
signif-icantly effect the cooking yield values of beef patties (p > 0.05). NAVEENAet al. (2008) found that the cooking yield values didn’t effect
from adding pomegranate juice, pomegranate rind powder extract and BHT as antioxidants in cooked chicken patties. Similarly to this study, SERDAROĞLUet al. (2007) reported that cooking loss of treated with citric
acid and grapefruit juice meat samples ranged from 22.4 to 33.3%. The cooking loss of 100% grapefruit juice was lower than other groups. NADZIRAHet al. (2016) recorded that the cooking loss value of
bromelain-treated beef was higher than untreated meat sample.
TBA and antioxidant activity
Table 2 shows that the the effect of fruit juice marination on the TBA value and antioxidant activity of chicken and turkey breast meat samples (p < 0.01). Treatment with juices had significantly (p < 0.01) higher levels of antioxidant activ-ity than control group. PJ showed a significantly higher antioxidant activity than control group. Pome-granate juice has been demon-strated to be high in antioxidant activity and is effective in preven-tion of atherosclerosis, low-density lipoprotein oxidation, prostate cancer, platelet aggre-gation and various cardiovascular diseases (ADHAMIand MUKHTAR,
2006). NAVEENAet al. (2008) have
found pomegranate juice had antioxidant activity in chicken meat patties.
....
....
...
...
...
...
...
...
...
....
....
....
....
....
....
.
Tab. 2: TBA values and antioxidant activity of
marinated chicken and turkey breast meat samples
Tab. 2: TBA-Werte und antioxidative Aktivität von mariniertem Hühner- und Putenbrustproben
Samples TBA DPPH Chicken breast C 0.30±0.01a 6.47±0.15c GJ 0.24±0.00b 9.46±0.36bc MJ 0.13±0.00d 11.91±0.14b PJ 0.15±0.01c 28.40±3.36a Turkey breast C 0.49a±0.01a 1.98±0.30c GJ 0.35±0.01b 3.76±0.44b MJ 0.15d±0.00d 5.07±0.52b PJ 0.26c±0.01c 15.44±0.84a
Values correspond to mean data ± corresponds to standard deviation. a–dWith different letters in columns are significantly different (p < 0.01).
C: Control (Distilled water), GJ: Marination with black grape juice, MJ: Marination with black mulberry juice, PJ: Marination with pomegranate juice
Source: ÜNALet al. FLEISCHWIRTSCHAFT 2_2020
TBA and antioxidant activity
....
....
....
....
....
....
....
...
...
...
...
...
...
...
...
Tab. 3: Average MORSF and MORSE values of chicken
and turkey breast samples
Tab. 3: Durchschnittliche MORSF- und MORSE-Werte von Hühner- und Putenbrustproben
Samples MORSF (N) MORSE (N.mm)
Chicken breast C 8.78±0.21 54.78±3.10 GJ 8.26±1.27 56.28±3.47 MJ 8.66±1.84 56.74±11.31 PJ 7.41±1.92 47.08±14.77 Turkey breast C 7.42±0.34a 41.89±5.31 GJ 5.88±0.05c 41.60±0.86 MJ 6.86±0.04b 42.35±0.65 PJ 7.49±0.26a 45.60±6.01
Values correspond to mean data ± corresponds to standard error. a–cWith different letters in columns are significantly different (p < 0.05).
C: Control (Distilled water), GJ: Marination with black grape juice, MJ: Marination with black mulberry juice, PJ: Marination with pomegranate juice
Source: ÜNALet al. FLEISCHWIRTSCHAFT 2_2020
MORSF and MORSE values
....
....
...
...
...
...
...
...
....
....
...
....
....
..
Tab. 4: Average
L*, a* and b* values of chicken and turkey breast samples
Tab. 4: Durchschnittliche L*-, a*- und b*-Werte von Hühner- und Putenbrustproben
Samples L* a* b* Chicken breast C 45.65c±0.04 4.46c±0.19 4.95b±1.13 GJ 47.99b±0.34 8.32b±0.32 2.64b±0.31 MJ 37.66d±0.24 15.53a±0.02 3.17b±0.08 PJ 51.21a±0.48 9.92b±0.76 16.01a±0.30 Turkey breast C 46.35a±0.28 6.42c±0.53 3.06b±0.13 GJ 43.73b±0.39 11.01b±0.11 2.09b±0.43 MJ 36.85c±0.55 16.01a±2.02 3.87b±0.54 PJ 43.42b±0.02 11.80ab±0.64 14.39a±1.06
Values correspond to mean data ± corresponds to standard deviation.a–dWith different letters in columns are significantly different (p < 0.01). C: Control (Distilled water), GJ: Marination with black grape juice, MJ: Marination with black mulberry juice,
PJ: Marination with pomegranate juice
Source: ÜNALet al. FLEISCHWIRTSCHAFT 2_2020
Forschung und Entwicklung
All the treatments significantly (p < 0.01) decreased the TBA values througout marination process compared to control. The increase in TBA values in turkey breast meat (0.49) was higher than chicken breast meat (0.26). The lipid oxidation inhibition effect was highest (p < 0.01) in MJ compared to control samples. The TBA values significantly ”(p < 0.01) were reduced in MJ treated with chicken and turkey meat samples. XUet al. (2018) reported that mulberry polyphenol
signifi-cantly decraesed the TBARS values of dried minced pork slices. VAITHIYANATHANet al. (2011) stated pomegranate fruit juice could delay
the TBARS values in chicken samples at 4 °C for 28 days.
Fruits serve as a significant source of antioxidant such as phenolic acids, and flavonoids. For example, MJ is showed to be a very concen-trated source of polyphenols and anthocyanins (SUHet al., 2003).
ESPADA-BELLIDOet al. (2017) reported that its antioxidant feature
origi-nated from phenolic acids, flavonols and anthocyanins contents. PJ contains high antioxidant activity, directly related to its phenolic content (VÁZQUEZ-ARAÚJOet al., 2011). ZHUet al. (2019) explained the
dominant phenolic compounds of grape pomace were phenolic acids, catechins, quercetin and its derivatives (flavanols), and anthocyani-dins. Phenolics has reacted with free radicals to convert them to more stable products and terminated free radical chain reactions (NEGIand
JAYAPRAKASHA, 2003). JONGBERGet al. (2013) reported antioxidants had
believed to break free radical chains of oxidation by donation of a hydrogen atoms from the phenolic groups, thereby preventing lipid or protein oxidation.
The best effect of lipid oxidation prevention has been showed in the MJ group, when the highest antioxidant activity was determined in PJ group. It is probably thought to be caused by the structure of the polyphenols makes a significant contribution to their antioxidant capacity. ZHUet al. (2019) stated that the number and position of
hy-droxyl groups in the phenolic molecule, which have effects on their
hydrogen atom denoting abilities and unpaired electron supporting capacities, will directly affect its antioxidant capacity.
Texture analysis
Average MORSF and MORSE values of chicken and turkey breast samples are given in Table 3. Marination treatments did not affect the MORSE values of the samples and also MORSF values of the marinated chicken breast samples (p > 0.05). However, the differences between MORSF values of marinated turkey samples were statistically significant (p < 0.05). Figure 1 indicates the effects of marination treatments on MORSF values of turkey breast samples.
While the group of C and PJ presented the highest MORSF values in turkey breasts, GJ had the lowest MORSF values. Marination with MJ increased tenderness of turkey breasts when compared to C and PJ. The increase in tenderness of marinated turkey breast samples may depend on the pH decline arising from acidic marinades. On the other hand, in acidic marinades, meat structure weakens and enhanced transformation of collagen to gelatine throughout the cooking process (ÖNENÇet al., 2004). GÖKand BOR(2016) studied the effects of marination
with fruit and vegetable juice on turkey breast meat tenderness. It was concluded from their study that samples marinated in pomegranate and red grape juices were significantly more tender than control and samples marinated with mixed vegetable, black mulberry, black carrot juices.
The results of grape juice on tenderness of turkey breast meat are similar to their study. However, the effect of PJ on tenderness of tur-key breast meat was not observed in this study. This difference be-tween this study and GÖKand BOR(2016) is probably due to the different
turkeys (ages, weights) used, differences in marinades (fruit juices) and marination conditions (duration, temperature, treatment of mari-nation, etc).
....
...
...
...
...
...
...
...
...
....
...
....
....
....
....
....
....
.
Quelle: ÜNALet el. FLEISCHWIRTSCHAFT 2_2020
Fig. 1: MORS diagrams of turkey breast samples (C: Control [distilled water], GJ: Marination with black grape juice, MJ: Marination with black mulberry juice, PJ: Marination with pomegranate juice)
Abb. 1: MORS-Diagramme von Putenbrustproben (C: Kontrolle [destilliertes Wasser], GJ: Marinieren mit schwarzem Traubensaft, MJ: Marinieren mit schwarzem Maulbeersaft, PJ: Marinieren mit Granatapfelsaft)
Colour properties
Colour is the first effective characteristic on the consumer preference and played an important role in meat and meat products quality (XUet
al., 2018). Table 4 shows L*, a* and b* values of chicken and turkey meat samples marinated with distilled water (control), GJ, MJ and PJ. In this study, treatment fruit juice had a significant (p < 0.01) effect on the colour values of meat samples. The chicken meat sample treated with PJ had the highest L* value (51.21) and also the meat sample treated with GJ had higher L* value (47.99) than the control and MJ treated samples (45.65 and 37.66, respectively). The chicken and turkey meat samples treated with MJ had the highest a* values (15.53, 16.01 re-spectively). Additionally, treating with PJ increased the b* values of chicken and turkey meat samples. But there was no significant differ-ences between the b* values of control and other samples.
SERDAROĞLUet al. (2007) recorded thatL* values of turkey meat
sam-ples increased and the a* values were not affected by marination in grapefruit juice and citric acid. KURT(2017) recorded that L* and b*
values of samples decreased as the addition of pomegranate seed flour. It was reported that the differences in the colour values were depend on the colour pigments content of pomegranate seed flour and anthocyanins affected importantly the colour of the pomegranate seeds. The differences in the studies might attributed to the difference in the composition and content of antioxidants (XUet al., 2018).
XUet al. (2018) notified thatL* values were much lower in the dried
minced pork slices added mulberry polyphenols than the other sam-ples. The results of mulberry polyphenols effects on the L* values are similar this study. The authors attributed that this result was could be due to the phenolic compounds in mulberry polyphenols. The fruit colour directly indicated the anthocyanin content of fruits (ARAMWITet
al., 2010). XUet al. (2018) reported that purple-coloured mulberry fruits
included generally the highest anthocyanin quantity pursued by pur-ple-red- and red-coloured fruits, respectively. CHENGet al. (2018) stated
that concentrated MJ, which had high anthocyanin content, increased the redness of the dried minced pork slices before baking and caused the meat slices had a distinctive purple colour due to the anthocyanin pigments. NADZIRAHet al. (2016) reported that theL* and b* values of
bromelin-treated beef were increased and a* value was decreased.
Sensory evaluation
Figure 2 indicates the appearance, texture, flavour and overall accep-tance of the marinated chicken and turkey breast samples. The ap-pearance results showed meat samples containing dark fruit juices had higher scores than unmarinated samples. The other sensory scores had close values between marinated and unmarinated samples. No spices and salt were added in the samples to eliminate distraction. All the samples were equally acceptable as evidenced by the sensory evalua-tion scores in accordance with statistically (p > 0.05). SERDAROĞLUet al.
(2007) found the appearance scores of breast meat samples decreased when treated with acidic marinades. NAVEENAet al. (2008) stated colour,
off-odour, sweet flavour, chicken flavour and overall palatability sen-sory evaluation scores did not reveal any significant difference in appearance between control and marinated samples.
Conclusions
The water holding properties andL*, a* and b* values of chicken and turkey meat samples were affected (p < 0.01) by marination with fruit juice. While the textural properties of chicken breast samples were not affected by the marination treatment, it affected the tenderness of turkey meat positively. Turkey breast samples marinated with black grape juice were determined to be the most tender. Marinating with natural fruit juices significantly inhibited lipid oxidation in chicken and turkey meats to a much greater extent than unmarinated group. Using fruit juices to marination increased (p < 0.01) the radical scavenging activity compared to control group. Further studies are needed to investigate natural agents in marination treatment in meat samples. References
1. ADHAMI, V.M. and MUKHTAR, H. (2006): Polyphenols from green tea and pomegtanate for
prevention of prostate cancer. Free Radical Research 40, 1095–1104. – 2. ALVARADO, C.
and MCKEE, S. (2007): Marination to improve functional properties and safety of poultry
meat. Journal of Applied Poultry Research 16, 113–120. – 3. ANANG, D.M., RUSUL, B., HOOI
LING, F. and BHAT, R. (2010): Inhibitory effects of lactic acid and lauricidin on spoilage
organisms of chicken breast during storage at chilled temperature. International Journal of Food Microbiology 144, 152–159. – 4. AOAC (2000): Official methods of
analy-....
...
...
...
...
...
...
...
....
....
....
....
....
Quelle: ÜNALet al. FLEISCHWIRTSCHAFT 2_2020
Fig. 2: Average values of consumer sensory attributes of chicken and turkey breast samples (C: Control [distilled water], GJ: Marination with black grape juice, MJ: Marination with black mulberry juice, PJ: Marination with pomegranate juice)
Abb. 2: Durchschnittswerte der sensorischen Verbrauchereigenschaften von Hühner- und Putenbrustproben (C: Kontrolle [destilliertes Wasser], GJ: Marinieren mit schwarzem Traubensaft, MJ: Marinieren mit schwarzem Maulbeersaft, PJ: Marinieren mit Granatapfelsaft)
sis. Assn. of Official Analytical Chemists., Washington, D.C. – 5. ARAMWIT, P., BANG, N. and
SRICHANA, T. (2010): The properties and stability of anthocyanins in mulberry fruits. Food
Research International 43, 1093–1097. – 6. BRAND-WILLIAMS, W., CUVEIER, M.E. and BERSET,
C. (1995): Use of a free radical method to evaluate antioxidant activity. LWT-Food Science and Technology 28, 25–30. – 7. CAVITT, L., YOUM, G., MEULLENET, J., OWENS, C. and
XIONG, R. (2004): Prediction of Poultry Meat Tenderness Using Razor Blade Shear,
Allo-Kramer Shear, and Sarcomere Length. Journal of Food Science and Technology 69, 11–15. – 8. CHENG, J.R., LIU, X.M., CHEN, Z.Y., ZHANG, Y.S. and ZHANG, Y.H. (2016): Mulberry
anthocyanin biotransformation by intestinal probiotics. Food Chemistry 213, 721–727. – 9. CHENG, J.R., LIU, X.M., ZHANG, W., CHEN, Z.Y. and WANG, X.P. (2018): Stability of phenolic
compounds and antioxidant capacity of concentrated mulberry juice-enriched dried-minced pork slices during preparation and storage Food Control 89, 187–195. – 10. DEVATKAL, S.K., Narsaiah, K. and BORAH, A. (2010): Anti-oxidant effect of extracts of
kinnow rind, pomegranate rind and seed powders in cooked goat meat patties. Meat Science 85, 155–159. – 11. ESPADA-BELLIDO, E., FERREIRO-GONZÁLEZ, M., CARRERA, C., PALMA, M.,
BARROSO, C.G. and BARBERO, G.F. (2017): Optimization of the ultrasound-assisted
extrac-tion of anthocyanins and total phenolic compounds in mulberry (Morus nigra) pulp. Food Chemistry 219, 23–32. – 12. FAO (2017): OECD-FAO Agrıcultural Outlook 2017 – 2026. – 13. FAO (2018): World Food Outlook. – 14. FAZAELI, M., HOJJATPANAH, G. and EMAM
-DJOMEH, Z. (2013): Effects of heating method and conditions on the evaporation rate and
quality attributes of black mulberry (Morus nigra) juice concentrate. Journal of Food Science and Technology 50, 35–43. – 15. FIRUZI, M.R., NIAKOUSARI, M., ESKANDARI, M.H.,
KERAMAT, M., GAHRUIE, H.H. and KHANEGHAH, A.M. (2019): Incorporation of pomegranate juice
concentrate and pomegranate rind powder extract to improve the oxidative stability of frankfurter during refrigerated storage. LWT-Food Science and Technology 102, 237–245. – 16. FULEKI, T. and RICARDO-DA-SILVA, M.J. (2003): Effects of cultivar and
pro-cessing method on the contents of catechins and procyanidins in grape juice. Journal of Agriculture and Food Chemistry 51, 640–646. – 17. GÖK, V. and BOR, Y. (2016): Effect of
marination with fruit and vegetable juice on the some quality characteristics of turkey breast meat. Brazilian Journal of Poultry Science 18, 481–488. – 18. HAN, Q., GAO, H., CHEN,
H., FANG, X. and WU, W. (2017): Precooling and ozone treatments affects postharvest
quality of black mulberry (Morus nigra) fruits. Food Chemistry 221, 1947–1953. – 19. HUNT, M., ACTON, J., BENEDICT, R., CALKINS, C., CORNFORTH, D., JEREMIAH, L., OLSON, D., SALM, C.,
SAVELL, J. and SHIVAS, S. (1991): Guidelines for Meat Color Evaluation American Meat
Science Association, Kansas State University, Manhattan. – 20. JONGBERG, S., TØRNGREN,
M.A., GUNVIG, A., SKIBSTED, L.H. and LUND, M.N. (2013): Effect of green tea or rosemary
extract on protein oxidation in Bologna type sausages prepared from oxidatively stressed pork. Meat Science 93, 538–546. – 21. KARGIOTOU, C., KATSANIDIS, E., RHOADES, J.,
KONTOMINAS, M. and KOUTSOUMANIS, K. (2011): Efficacies of soy sauce and wine base
mari-nades for controlling spoilage of raw beef. Food Microbiolology 28, 158–163. – 22. KONDAIAH, N., ANJANEYULU, A., RAO, V.K., SHARMA, N. and JOSHI, H. (1985): Effect of Salt and
Phosphate on the Quality of Buffalo and Goat Meats. Meat Science 15, 183–192. – 23. KURT, Ş. (2017): Investigation of the Potential Usage of Pomegranate Seed Flour in
the Beef Patties. Adıyaman University Journal of Science 7, 1–11. – 24. LEE, Y. and HWANG,
K.T. (2017): Changes in physicochemical properties of mulberry fruits (Morus alba L.) during ripening. Scientia Horticulturae 217, 189–196. – 25. LYTOU, A.E., PANAGOU, E.Z. and
NYCHAS, G.-J.E. (2017): Effect of different marinating conditions on the evolution of
spoilage microbiota and metabolomic profile of chicken breast fillets. Food Microbiolol-ogy 66, 141–149. – 26. NADZIRAH, K., ZAINAL, S., NORIHAM, A. and NORMAH, I. (2016): Application
of Bromelain Powder Produced from Pineapple Crowns in Tenderising Beef Round Cuts. International Food Research Journal 23, 1590–1599. – 27. NAVEENA, B.M., SEN, A.R.,
VAITHIYANATHAN, S., BABJI, Y. and KONDAIAH, N. (2008): Comparative efficacy of pomegranate
juice, pomegranate rind powder extract and BHT as antioxidants in cooked chicken patties. Meat Science 80, 1304–1308. – 28. NEGI, P.S. and JAYAPRAKASHA, G.K. (2003):
Antioxidant and antibacterial activities of Punica granitum peel extracts. Journal of Food Science 67, 1473–1477. – 29. NISIOTOU, A., CHORIANOPOULOS, N.G., GOUNADAKI, A., PANAGOU,
E.Z. and NYCHAS, G.J.E. (2013): Effect of wine-based marinades on the behavior of
Salmo-nella typhimurium and background flora in beef fillets. International Journal of Food Microbiology 164, 119–127. – 30. OCKERMAN, H.W. (1985): Quality control of post-mortem
muscle tissue. The Ohio State University, Colombus, OH. – 31. ÖNENÇ, A., SERDAROĞLU, M.
and ABDRAIMOV, K. (2004): Effect of various additives to marinating baths on some
properties of cattle meat. European Food Research and Technology 218, 114–117. – 32. PARK, Y.K., PARK, E., KIMA, J.-S. and KANG, M.-H. (2003): Daily grape juice consumption
reduces oxidative DNA damage and plasma free radical levels in healthy Koreans. Mutation Research 529, 77–86. – 33. PATHANIA, A., MCKEE, S.R., BILGILI, S.F. and SINGH, M.
(2010): Antimicrobial activity of commercial marinades against multiple strains of Salmonella spp. International Journal of Food Microbiology 139, 214–217. – 34. POKORNY,
J. (1991): Natural antioxidants for food use. Trends in Food Science and Technology 2, 223–227. – 35. RADHAKRISHNAN, K., BABUSKIN, S., AZHAGUSARAVANABABU, P., SASIKALA, M.,
SABINA, K., ARCHANA, G., SIVARAJAN, M. and SUKUMAR, M. (2014): Antimicrobial and
antioxi-dant effects of spice extracts on the shelf life extension of raw chicken meat. Interna-tional Journal of Food Microbiology 171, 32–40. – 36. SAHA, A., LEE, Y., MEULLENET, J. and
OWENS, C. (2009): Consumer Acceptance of Broiler Breast Fillets Marinated with Varying
Levels of Salt. Poultry Science 88, 415–423. – 37. SERDAROĞLU, M., ABDRAIMOV, K. and ÖNENÇ,
A. (2007): The Effects of Marinating with Citric Acid Solutions and Grapefruit Juice on Cooking and Eating Quality of Turkey Breast. Journal of muscle foods 18, 162–172. – 38. SLAVIN, M., LU, Y., KAPLAN, N. and YU, L. (2013): Effects of baking on
cyanidin-3-gluco-side content and antioxidant properties of black and yellow soybean crackers. Food Chemistry 141, 1166–1174. – 39. SUH, H.J., NOH, D.O., KANG, C.S., KIM, J.M. and LEE, S.W.
(2003): Thermal kinetics of color degradation of mulberry fruit extract. Food Nahrung 47, 132–135. – 40. VAITHIYANATHAN, S., NAVEENA, B.M., MUTHUKUMAR, M., GIRISH, P.S. and KONDAIAH, N.
(2011): Effect of dipping in pomegranate (Punica granatum ) fruit juice phenolic solution on the shelf life of chicken meat under refrigerated storage (4 °C). Meat Science 88, 409–414. – 41. VÁZQUEZ-ARAÚJO, L., CHAMBERSIV, E., ADHIKARI, K. and CARBONELL-BARRACHINA,
A.A. (2011): Physico-chemical and sensory properties of pomegranate juices with pomegranate albedo and carpellar membranes homogenate. LWT-Food Science and Technology 44, 2119–2125. – 42. WARDLAW, F.B., SKELLEY, G.C., JOHNSON, M.G. and ACTON, J.C.
(1973): Changes in meat components during fermentation, heat procesing and drying of a summer sausage. Journal of Food Science 38, 1128–1231. – 43. WHITE, H. and KOKOTSAKI,
K. (2004): Indian food in the UK: personal values and changing patterns of consumption. International Journal of Consumer Studies 28, 284–294. – 44. XU, L., ZHU, M.J., LIU, X.M.
and CHENG, J.R. (2018): Inhibitory effect of mulberry (Morus alba) polyphenol on the lipid
and protein oxidation of dried minced pork slices during heat processing and storage. Lebensmittel-Wissenschaft und-Technologie-Food Science and Technology 91, 222–228. – 45. YUSOP, S.M. (2011): Marinating and enhancement of the nutritional
content of processed meat products. In: J.P. Kerry and K. J.F. (Eds.), Processed Meats 17, 421–449. – 46. ZHU, M., HUANG, Y., WANG, Y., SHI, T., ZHANG, L., CHEN, Y. and XIE, M. (2019):
Comparison of (poly) phenolic compounds and antioxidant properties of pomace extracts from kiwi and grape juice. Food Chemistry 271, 425–432.
Zusammenfassung
Technologische und strukturelle Eigenschaften von mit
verschiedenen dunklen Fruchtsäften marinierten
Hühner-und Putenbrustfilets
By Kübra Ünal, Halime Alp, A. Samet Babaoğlu, and Mustafa Karakaya – Konya/Türkei
Brustfleisch | Dunkle Farbe | Fruchtsäfte | Marinieren | Textur
In dieser Untersuchung wurden die Auswirkungen dreier verschiedener dunkel gefärbter Marinaden mit schwarzem Maulbeer- (MJ), Trauben- (GJ) und Granatapfelsaft (PJ) auf den pH-Wert, das Wasserhaltevermögen (WHC), den Kochverlust (CL) und die Farbe (L*, a*, b*) von Hühner- und Putenbrustfleisch ermittelt. Außerdem wurden die Lipidoxidation (Thio-barbitursäurezahl-TBA), die antioxidative Aktivität (DPPH-2,2-Diphenyl-1-Picrylhydrazyl), die texturalen Eigenschaften (Meullenet-Owens Scherkraft und Energie) und sensorische Analysen der Proben durchge-führt. Die Fruchtsäfte senkten im Allgemeinen die pH- und WHC-Werte im Vergleich zur Kontrolle. Die mit MJ behandelten Fleischproben hatten den höchsten a*-Wert und die Marinierung mit PJ erhöhte die b*-Werte der Proben. Die mit GJ marinierten Putenbrustproben wurden als die zartesten bestimmt. Die Marinierung mit Fruchtsäften hemmte die Lipidoxidation signifikant und erhöhte (p < 0,01) die DPPH-Werte in Hühner- und Puten-fleisch. Diese natürlichen Marinaden sollten empfohlen werden, um die Verbraucherakzeptanz von Hühner- und Putenbrustfleisch zu entwickeln.
Authors’ addresses
Kübra Ünal (Correspondence author: [email protected]), A. Samet Babaoğlu , Mustafa Karakaya, Food Engineering Department, Agricultural Faculty, Selcuk University, Konya 42050, Turkey; Halime Alp, Department of Food Processing, Karapınar Aydoğanlar Vocational School, Selcuk University, 42400, Karapınar/Konya, Turkey
Forschung und Entwicklung
View publication stats View publication stats

![Fig. 1: MORS diagrams of turkey breast samples (C: Control [distilled water], GJ: Marination with black grape juice, MJ: Marination with black mulberry juice, PJ: Marination with pomegranate juice)](https://thumb-eu.123doks.com/thumbv2/9libnet/4972242.100657/5.977.67.925.138.610/diagrams-control-distilled-marination-marination-mulberry-marination-pomegranate.webp)
