Background: There are a lot study confirmed the relationship of bone serum markers changes and skeletal irregularities. But there is no sufficient case control studies about the role of these markers on bisphosphonate induced osteonecrosis of jaws (BRONJ). Aims: The aim of this study is to find out if there is any derangement of bone markers in bisphosphonate‑treated patients with ONJ. Methods: We obtained serum bone markers and other relevant endocrine assays on 20 patients with osteonecrosis of the jaw (ONJ) and 20 randomized healthy volunteers. All of the ONJ group treated with zoledronic acid and had been withdrawn from bisphosphonate for at least 6 months. Diagnostic criteria for ONJ were those formulated by the American Association of Oral and Maxillofacial Surgeons. Serum levels of several indices of bone remodeling were evaluated using commercial enzyme‑linked immunosorbent assays. The biochemical assays were performed on N‑Telopeptides of type I collagen (NTX), bone‑specific alkaline phosphatase (ALP), calcitonin, osteocalcin, intact parathyroid hormone (PTH), T3, T4, TSH, and Vitamin D 25 hydroxy (Vit‑D). Results: In ONJ group, PTH level is statistically higher and TSH, Vit‑D, osteocalcin and NTX levels statistically lower compared to control group. Conclusion: We conclude that these changes in PTH, Vit‑D, TSH, osteocalcin and NTX levels maybe have a role in the pathophysiology of BRONJ. But the data need to be confirmed by future studies.
Keywords: Bisphosphonate, bone markers, bone metabolism, oral pathology, osteonecrosis
Changes in Serological Bone Turnover Markers in Bisphosphonate
Induced Osteonecrosis of the Jaws: A Case Control Study
S Demircan, SC Işler1
Address for correspondence: Dr. S Demircan, Beykent University Vocational School, Dental Prosthetic Technology, Istanbul, Turkey. E‑mail: [email protected] condition of osteonecrosis of the jaw and facial bones.[1,2]
The mechanism that underlies the association between intravenous bisphosphonate treatment and jaw osteonecrosis is not understood. Current explanations include an infectious etiology, the loss of blood supply or the suppression of bone turnover.[3‑5] There is also a
lack of information on the epidemiology of the toxicity. There is no information from prospective trials and only limited information from observational cohorts on incidence, time course, or other risk factors.[6,7]
Introduction
B
isphosphonates have been used in the United States (USA) to treat osteoporosis since 1977. Oral and intravenous bisphosphonates are the preferred pharmacologic agents in the treatment of osteoporosis. Highly potent intravenously administered nitrogen‑containing bisphosphonates were introduced for the treatment of cancer‑related bone lesions and hypercalcemia in 1991 and in 2001. Intravenous bisphosphonates are also effective in preventing osteopenia in patients with prostate cancer undergoing androgen deprivation therapy. Starting in 2003, a growing number of case reports and case series have been published that link bisphosphonate administration, particularly bisphosphonates administered intravenously with the previously rareDepartment of Dental Prosthetic Technology, Beykent University Vocational School,
1Department of Oral Surgery,
İstanbul University Faculty of Dentistry, Beykent, Istanbul, Turkey
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution‑NonCommercial‑ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non‑commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
For reprints contact: [email protected]
How to cite this article: Demircan S, Işler SC. Changes in serological bone turnover markers in bisphosphonate induced osteonecrosis of the jaws: A case control study. Niger J Clin Pract 2020;23:154-8.
Access this article online
Quick Response Code:
Website: www.njcponline.com DOI: 10.4103/njcp.njcp_374_19 PMID: *******
Abstract
Received: 18-Jul-2019; Revision: 19-Aug-2019; Accepted: 10-Sep-2019; Published: 06-Feb-2020154 Nigerian Journal of Clinical Practice ¦ Volume 23 ¦ Issue 2 ¦ February 2020 Nigerian Journal of Clinical Practice ¦ Volume 23 ¦ Issue 2 ¦ February 2020 155 Recent studies have shown that there are strong
correlations between levels of bone markers and the occurrence of skeletal morbidity, confirming earlier small case series from a variety of centers.[8‑11] Assessment of
these markers provides specific insight into rates of bone formation and resorption and levels seem to correlate with bone pain levels and the burden of disease in the skeleton.[11,12] But there is no efficient case control studies
about the role of these markers on bisphosphonates induced osteonecrosis of the jaw (BRONJ).
In this study, we aimed to determine if there is a relationship between osteonecrosis of the jaws by patients treated with intravenous bisphosphonates and changes in serum bone markers.
The study approved by ethics committee of Cerrahpaşa Medical Faculty of the University of Istanbul. Date 09.2008.
Methods
Considering the recent literature we select PTH, T3, T4, TSH, and Vit‑D, calcitonin, osteocalcin, ALP and NTX values for evaluation. We obtained serum bone markers and other relevant endocrine assays on 20 patients with BRONJ (Study Group) and 20 randomized healthy volunteers (Control Group).
Diagnostic criteria for study group were those formulated by the American Association of Oral and Maxillofacial Surgeons. Exclusion criteria included known metabolic bone disease other than bone metastases or osteoporosis, treatment with bisphosphonates within 6 months prior to randomization, or other treatments with effects on bone, e.g. calcitonin, fluoride, and hormone replacement therapy, within 6 months prior to randomization. The exclusion criteria also included local radiotherapy within 2 weeks prior to randomization, hypercalcemia, impaired renal function, liver dysfunction, life expectancy of less than 3 months, pregnancy, or lactation. Local field radiotherapy was allowed during the study. All patients were postmenopausal.
12 of our patients in ONJ group were women. All of them had metastatic breast cancer and had been treated with zoledronic acid. 8 men patients in ONJ group had metastatic prostate cancer treated with zoledronic acid. All patients had been treated with zoledronic acid for 18 months and had been withdrawn from bisphosphonate for at least 6 months. None were taking or had taken corticosteroids.
Our control group had been consist of 12 female and 8 male healthy volunteers to be compatible with the study group. Before study all patients informed about the study and written consents taken by the authors.
This prospective study was reviewed and approved by the Institutional Review Board of the Istanbul University Faculty of Medicine.
Biochemical parameters
Overnight‑fasting blood samples were taken in the morning from the antecubital vein. The blood samples were centrifuged at 2500 × g for 15 minutes. T3, T4, TSH and PTH were analyzed using chemiluminescent microparticle immunoassay (CMIA) on the Abbott Architect i2000 immunoassay analyzer (Abbott Laboratories, Abbott Park, IL, USA) in human serum. ALP was analyzed using the para‑Nitrophenyl‑phosphate (p‑NPP) methods by Abbott Architect c8000 immunoassay analyzer (Abbott Laboratories, Abbott Park, IL). Calcitonin was measured with Immulite 2000 analyzer (DPC, USA) by chemiluminescent immunometric assay.
Serum Osteocalcin was assayed with a two‑site immunochemiluminometric assay adapted on the Liaison (Sangtec, Germany) automated analyzer, Plasma level of 25‑OH VitaminD3 was determined by high performance liquid chromatography (HPLC) (ImmuChrom, Bensheim, Germany).
NTX levels in serum were measured with competitive inhibition enzyme‑linked immunosorbent assay (ELISA/ EIA) (Osteomark, Wampole Laboratories, USA).
Statistical analysis
Comparisons between groups were made by Mann‑Whitney U test. Correlation between variables were tested by Chi‑squared test. NCSS (2007) was used for statistical analysis; P value lower than 0.05 was considered significant.
Results
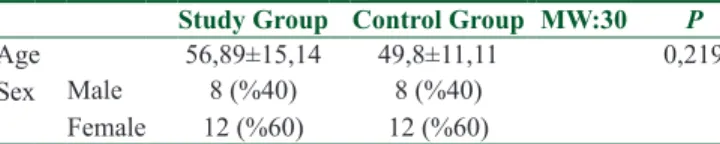
There was no significant differences for age and sex distribution [Table 1]. Bone markers and other relevant endocrine assays on 20 patients with BRONJ and 20 randomized healthy volunteers shown in Table 2.
No significant difference was seen in T3 (P = 0.165), T4 (P = 0.253), ALP (P = 0.744) and calcitonin (P = 0.252) levels between two groups.
In study group, the concentrations of serum PTH level statistically higher than control group (P = 0.009). The concentrations of serum osteocalcin level statistically
Table 1: Age and sex distribution of study and control groups
Study Group Control Group MW:30 P
Age 56,89±15,14 49,8±11,11 0,219
Sex Male 8 (%40) 8 (%40)
lower than the control group (P = 0.014). The concentrations of NTX level statistically lower than the control group (P = 0.003). The concentrations of TSH level statistically lower than the control group (P = 0.027). The concentrations of Vit‑D level statistically lower than the control group (P = 0.046).
Discussion
The measurement of biochemical markers of bone metabolism has become more common in recent trials in patients with bone metastases. Assessment of these markers provides specific insight into rates of bone formation and resorption and levels seem to correlate with bone pain levels and the burden of disease in the skeleton, while being unaffected by the burden of soft tissue or visceral disease.[8‑10] They are signs of the bone
turnover process and may be used as an aid to bone mineral density testing when determining whether or not a patient might have a bone disease.[13‑15]
A number of other studies have been published studying bone turnover markers as a sign of bone diseases[16,17]
or progression.[13,14,18] Although one or more bone
marker assays may be ordered to help identify patients with increased bone resorption and/or formation rates, their routine role in clinical practice and correlation with bisphosphonate usage is unproven. Systemic effects of bisphosphonates include acute phase reaction and hypocalcaemia associated with the first infusion, increased BMD and BMC, increased cortical thickness and trabecular number.[19‑21] Bisphosphonates also result
in reduced bone turnover and altered bone modeling.[19,22]
Otto et al. did a multi‑center case control study and suggested that the relative frequency of osteoporosis patients on oral Bps suffering from BRONJ is higher than previously reported.[11]
Considering these publications, we aimed to determine if there is a relationship between osteonecrosis of the jaws by patients treated with intravenous bisphosphonates and changes in serum bone markers.
Lein and others studied 6 bone markers after patients started zoledronic acid treatment for metastatic bone disease. The data presented in that paper did not show a significant correlation between baseline values of the bone markers and progression, but did show a significant association between increased bone markers (not including CTx) over the course of the study and disease progression.[15] But there are case control studies shown
that Ctx is not a definitive predictor of the development of BRONJ.[12,23,24]
Our results show significant association between TSH, PTH, osteocalcin, NTx, Vit‑D levels and ONJ. Elevated PTH values suggest excessive suppression of bone turnover. We also find elevated PTH levels associated with the ONJ. In a large population recruited in the registration trials of ZA, hyperpara‑thyroidism after ZA administration has been recently associated with worse prognosis.[25] Lehrer et al. studied normal serum bone
markers in bisphosphonate‑induced osteonecrosis of the jaws. They reported bone markers were within the normal range, except for intact parathyroid hormone, which was slightly elevated in one case of metastatic breast cancer.[26] Ardine et al. have postulated that
long‑term persistence of low serum calcium levels and high serum parathyroid hormone levels during bisphosphonate treatment predispose metastatic breast cancer patients to undergo ONJ.[27] These data need to
be confirmed by larger studies.
In contrast with our findings, significantly higher NTX levels in patients with bone metastases was reported previously[16,22] But a recent publication suggested that
increased markers of osteoclastic activity in patients with prostate cancer and bone metastases might be due to the effects of androgen deprivation therapy alone, rather than an effect of metastatic disease on the bone.[17]
Thyroid hormones are essential for normal skeletal growth and the maintenance of bone mass in adulthood. In adult, T3 stimulates osteoplastic bone resorption. TSH seems to be an inhibitor of bone resorption and formation. Adults with suppressed TSH from any cause have an increased risk of osteoporotic fracture.[28] Direct
osteotropic effect of TSH has been supported by studies analyzing the influence of recombinant TSH on the skeleton.[29] Maybe in TSH have a major role in ONJ,
but these data need to be confirmed by larger studies. As osteocalcin is produced by osteoblasts, it is often used as a marker for the bone formation process. It has been observed that higher serum‑osteocalcin levels are relatively well correlated with increases in bone mineral density (BMD) during treatment with anabolic Table 2: Bone markers and other relevant endocrine
assays of study and control groups
Study Group Control Group MW P
T3 0,98±0,26 1,21±0,36 28 0,165 T4 9,24±2,49 8,17±2,72 31 0,253 TSH 1,17±0,96 2,09±1 18 0,027 ALP 67,78±22 77,7±39,83 41 0,744 PTH 80,92±24,92 56,92±7,46 13 0,009 Vit‑D 22,43±9,36 29,9±8,22 20,5 0,046 Calcitonin 4,09±3,37 6,08±3,49 31,5 0,252 Osteocalsin 14,21±6,81 24,45±8 15 0,014 NTX 6,08±3,66 12,31±2,58 9 0,003
156 Nigerian Journal of Clinical Practice ¦ Volume 23 ¦ Issue 2 ¦ February 2020 Nigerian Journal of Clinical Practice ¦ Volume 23 ¦ Issue 2 ¦ February 2020 157 bone formation drugs for osteoporosis. In many studies,
osteocalcin is used as a preliminary biomarker on the effectiveness of a given drug on bone formation.[30,31] We
observed suppressed osteocalcin levels in ONJ group and this have effect on bone turnover.
Vitamin D deficiency associated with increase in bone turnover and enhanced bone loss.[32] And there are several
reports that associate vitamin D deficiency and increased PTH values.[33,34] We found vitamin D deficiency and
increased PTH values. Maybe PTH and vitamin D levels must be evaluated separately in a well‑rounded study.
Conclusion
We concluded that there is a strong relationship between the changes in PTH, Vit‑D, TSH, osteocalcin and NTX levels and BRONJ. The clinician must follow up the changes in biochemical markers of bone metabolism in managing the oral surgery patients on BPs. There is a need for further studies to determine the exact roles of these markers in the pathophysiology of BRONJ.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Marx RE. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: A growing epidemic. J Oral Maxillofac Surg 2003;61:1115‑7.
2. Farrugia MC, Summerlin DJ, Krowiak E, Huntley T, Freeman S, Borrowdale R, et al. Osteonecrosis of the mandible or maxilla associated with the use of new generation bisphosphonates. Laryngoscope 2006;116:115‑20.
3. Hellstein JW, Marek CL. Bisphosphonate osteochemonecrosis (bis‑phossy jaw): Is this the phossy jaw of the 21st century. J Oral Maxillofac Surg 2005;63:682‑9.
4. Cheng A, Mavrokokki A, Carter G, Stein B, Fazzalari NL, Wilson DF, et al. The dental implications of bisphosphonates and bone disease. Aust Dent J 2005;50(Suppl 2):S4‑13.
5. Otto S, Schreyer C, Hafner S, Mast G, Ehrenfeld M, Storzenbaum S, et al. Bisphosphonate‑related osteonecrosis of the jaws‑Characteristics, risk factors, clinical features, localization and impact on oncological treatment. J Craniomaxillofac Surg 2012;40:303‑9.
6. Bamias A, Kastritis E, Bamia C, Moulopoulos LA,
Melakopoulos I, Bozas G. Osteonecrosis of the jaw in cancer after treatment with bisphosphonates: Incidence and risk factors. J Clin Oncol 2005;23:8580‑7.
7. Abu‑Id MH, Warnke PH, Gottschalk J, Springer I, Wiltfang J, Acil Y, et al. “Bis‑phossy jaws”‑high and low risk factors for bisphosphonate‑induced osteonecrosis of the jaw. J Craniomaxillofac Surg 2008;36:95‑103.
8. Vinholes JJ, Purohit OP, Abbey ME, Eastell R, Coleman RE. Relationships between biochemical and symptomatic response in a double‑blind randomised trial of pamidronate for metastatic bone disease. Ann Oncol 1997;8:1243‑50.
9. Berruti A, Dogliotti L, Gorzegno G, Torta M, Tampellini M, Tucci M, et al. Differential patterns of bone turnover in relation to bone pain and disease extent in bone in cancer patients with skeletal metastases. Clin Chem 1999;45:1240‑7.
10. Hoskin PJ, Stratford MR, Folkes LK, Regan J, Yarnold JR. Effect of local radiotherapy for bone pain on urinary markers of osteoclast activity. Lancet 2000;355:1428‑9.
11. Otto S, Abu‑Id MH, Fedele S, Warnke PH, Becker ST, Kolk A, et al. Osteoporosis and bisphosphonates‑related osteonecrosis of the jaw: Not just a sporadic coincidence‑‑A multi‑centre study. J Craniomaxillofac Surg 2011;39:272‑7.
12. Lazarovici TS, Mesilaty‑Gross S, Vered I, Pariente C, Kanety H, Givol N, et al. Serologic bone markers for predicting development of osteonecrosis of the jaw in patients receiving bisphosphonates. J Oral Maxillofac Surg 2010;68:2241‑7. 13. Tamada T, Sone T, Tomomitsu T, Jo Y, Tanaka H, Fukunaga M.
Biochemical markers for the detection of bone metastasis in patients with prostate cancer: Diagnostic efficacy and the effect of hormonal therapy. J Bone Miner Metab 2001;19:45‑51. 14. Scher H, Warren M, Heller G. The association between measures
of progression and survival in castrate‑metastatic prostate cancer. Clin Cancer Res 2007;13:1488‑92.
15. Lein M, Wirth M, Miller K, Eickenberg HU, Weissbach L, Schmidt K, et al. Serial markers of bone turnover in men with metastatic prostate cancer treated with zoledronic Acid for detection of bone metastases progression. Eur Urol 2007;52:1381‑7.
16. Garnero P, Buchs N, Zekri J, Rizzoli R, Coleman R, Delmas P. Markers of bone turnover for the management of patients with bone metastases from prostate cancer. Br J Cancer 2000;82:858‑64.
17. Jung K, Lein M, Stephan C, Von Hösslin K, Semjonow A, Sinha P, et al. Comparison of 10 serum bone turnover markers in prostate carcinoma patients with bone metastatic spread: Diagnostic and prognostic implications. Int J Cancer 2004;111:783‑91.
18. Johansen JS, Brasso K, Iversen P, Teisner B, Garnero P, Price P, et al. Changes of biochemical markers of bone turnover and YKL‑40 following hormonal treatment for metastatic prostate cancer are related to survival. J. Clin Cancer Res 2007;13:3244‑9. 19. Munns CF, Rauch F, Travers R, Glorieux FH. Effects of
intraveneous pamidronate treatment in infants with osteogenesis imperfecta: Clinical and histomorphometric outcome. J Bone Miner Res 2005;20:1235‑43.
20. Land C, Rauch F, Glorieux FH. Cyclical of intraveneous pamidronate treatment in growing patients with osteogenesis imperfecta. J Bone Miner Res 2006;21:374‑9.
21. Munns CF, Rajab MH, Hong J, Briody J, Högler W, McQuade M, et al. Acute phase response and mineral status following low dose intravenous zoledronic acid in children. Bone 2007;41:366‑70.
Mumm S. Bisphosphonate‑induced osteopetrosis. N Engl J Med 2003;349:457‑63.
23. Lee CY, Suzuki JB. CTX biochemical marker of bone metabolism. Is it a reliable predictor of bisphosphonate‑associated osteonecrosis of the jaws after surgery? Part I: Biological concepts with a review of the literature. Implant Dent 2009;18:492‑500.
24. Lee CY, Suzuki JB. CTX biochemical marker of bone metabolism. Is it a reliable predictor of bisphosphonate‑associated osteonecrosis of the jaws after surgery? Part II: A prospective clinical study. Implant Dent 2010;19:29‑38.
25. Cosman F, Eriksen EF, Recknor C, Miller PD, Guañabens N, Kasperk C, et al. Effects of intravenous zoledronic acid plus subcutaneous teriparatide [rhPTH(1‑34)] in postmenopausal osteoporosis. J Bone Miner Res. 2011;26:503‑11.
26. Lehrer S, Montazem A, Ramanathan L, Pessin‑Minsley M, Pfail J, Stock RG, et al. Normal serum bone markers in bisphosphonate‑induced osteonecrosis of the jaws. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2008;106:389‑91. 27. Ardine M, Generali D, Donadio M, Bonardi S, Scoletta M,
Vandone AM. Could the long‑term persistence of low serum calcium levels and high serum parathyroid hormone levels during bisphosphonate treatment predispose metastatic breast cancer patients to undergo osteonecrosis of the jaw? Ann Oncol 2006;17:1336‑7. 28. Galliford TM, Murphy E, Williams AJ, Bassett JH, Williams GR.
Rewiev effects of thyroid status on bone metabolism: A primary role for thyroid stimulating hormone or thyroid hormone?
Minerva Endocrinol 2005;30:237‑46.
29. Sampath TK, Simic P, Sendak R, Draca N, Bowe AE, O’Brien S, et al. Thyroid‑stimulating hormone restores bone volume, microarchitecture, and strength in aged ovariectomized rats. J Bone Miner Res 2007;22:849‑59.
30. Meyer U, Meyer T, Vosshans J, Joos U. Decreased expression of osteocalcin and osteonectin in relation to high strains and decreased mineralization in mandibular distraction osteogenesis. J Craniomaxillofac Surg 1999;27:222‑7.
31. Rodrigues Luvizuto E, Monica Dias S, Chiantelli Cloudio‑Coutinho C, Alves Machado L, Rangel Garcia‑Jonior I, Okamoto T, et al. Temporal localization of osteocalcin protein during healing of tooth extraction sockets in rats. Minerva Stomatol 2010;59:355‑61.
32. Sahota O, Mundey MK, San P, Godber IM, Lawson N, Hosking DJ. The relationship between vitamin D and parathyroid hormone: Calcium homeostasis, bone turnover, and bone mineral density in postmenopausal women with established osteoporosis. Bone 2004;35:312‑9.
33. Bettica P, Bevilacqua M, Vago T, Norbiato P. High prevalence of hypovitaminosis D among free‑living postmenopausal women referred to an osteoporosis outpatient clinic in Northern Italy for initial screening. Osteoporos Int 1999;9:226‑9.
34. Aguado P, Campo MT, Garces MV. Low vitamin D levels in outpatient postmenopausal women from a rheumatology clinic in Madrid, Spain: Their relationship with bone mineral density. Osteoporos Int 2000;11:739‑44.