See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/285549966
Lip leishmaniasis: Clinical characteristics of
621 patients
Article · October 2015 DOI: 10.4103/2229-5151.170849 CITATION1
READS34
6 authors, including: Some of the authors of this publication are also working on these related projects: A cese of erythema multiforme developing after levetiracetam therapyView project Modelling Insect-Egg Data with Excess Zeros Using Zero-inflated Regression Models.
View project Yavuz Yeşilova Special Lokman Physician Hospital, Van, Tur… 82 PUBLICATIONS 268 CITATIONS SEE PROFILE Ahmet Uluat birecik state hospital, şanliurfa, birecik 3 PUBLICATIONS 1 CITATION SEE PROFILE Nurittin Ardic Baskent University 53 PUBLICATIONS 334 CITATIONS SEE PROFILE Abdullah Yesilova University of Yuzuncu Yil VAN 28 PUBLICATIONS 31 CITATIONS SEE PROFILE
All content following this page was uploaded by Yavuz Yeşilova on 07 February 2016.
ISSN : 2229-5151
Vol 5 / Issue 4 / October-December 2015
Official Publication of International Network of
Critical Illness and Injury Trial Experts
www.ijciis.org
International
Journal of
Critical Illness
&
Injury Science
IJCIIS
JJJJJ
JJC
J
JJC
J
C
C
C
C
CII
CII S
C IIS
C
C S
C
C
C
C
IIII
I
II
IIS
S
S
S
S
S
S
S
S
S
S
S
S
S
S
IIIIJ
J
JJC
JJC
JJC
J
J
J
JJJJJ
C
C
C
C
C
C
C
CII
CIII
CII
C
C IIS
C
C
C
C
C
C
IIS
I
I
I
I
IIS
I
I
S
S
S
S
S
S
S
S
S
S
S
S
S
S
S
S
S
IIJJJ
IIIIJ
S
S
S
S
S
S
Connecting Cells, Clinics and Communities across Continents
International Journal of
Critical Illness and Injury Science •
V olume 5 • Issue 4 • October-December 2015 • Pages 223-000 Included Included
265 © 2015 International Journal of Critical Illness and Injury Science | Published by Wolters Kluwer - Medknow
Dear Editor,
Old world leishmaniasis is seen in a wide geographical area including Turkey, as well. The causative agent is generally Leishmaniasis tropica, and the agent is rarely identified to be leishmaniasis major in such patients.[1] Depending on the leishmaniasis type and
the person’s immune system, it may be in one of the cutaneous, mucocutaneous leishmaniasis (ML), or visceral leishmaniasis (VL) forms.[2] Mucosal involvement
is rarely seen with old world leishmaniasis.[3] ML
disease is an important endemic disease and public health problem in undeveloped countries, since it has a significant rate of morbidity and mortality.[4]
This study presents a retrospective assessment of 621 leishmaniasis patients with mucosal involvement. 14,400 leishmaniasis patients were retrospectively examined; after that 621 patients with lip mucosa involvement were assessed. One patient also had accompanying gingiva involvement. Only one of the patients with lip involvement had genital involvement, and this patient was excluded from the study. The patients are diagnosed using clinical and parasitological methods (direct examination by scraping or imprint, histopathology, and culture).
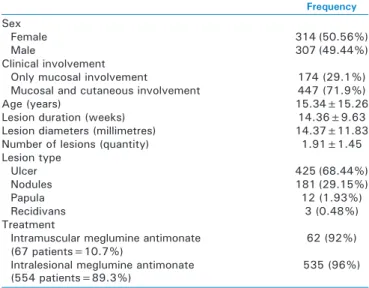
621 out of 14,400 leishmaniasis patients (4.3%) were identified to have mucosal involvement. 71.9% of patients with mucosal involvement (447 patients) also had accompanying cutaneous involvement. Female patients had the majority (F/M = 314/307), and their average age was 15.34 ± 15.26 years. The average lesion duration of the patients was identified as 14.36 ± 9 63 weeks. The lesion diameters and numbers were 14.37 ± 11.83 mm and 1.91 ± 1.45, respectively. As per the assessment made on the basis of lesion types, the ulcerated type lesions (68.44%) were the ones most frequently seen in leishmaniasis patients, followed by nodules (29.15%), papules (1.93%), and residence type 3 (0.48%) lesions, respectively. The ratio of patients receiving intramuscular injections of meglumine antimoniate was 10.7% (67 patients), and the treatment response rate was approximately 92%. As for the ratio of patients receiving intralesional injections of meglumine antimoniate, it was identified as 89.3% (554 patients) and the ratio of patients with treatment response was determined as 96% [Table 1]. While mucosal involvement is often seen with new world leishmaniasis, involvement is limited in the old
world leishmaniasis.[5‑7] An ML case without cutaneous
lesions reported in Sudan by Ibrahim et al. also had visceral involvement.[5] In addition, another ML case
reported in Sudan had involvement of the larynx, palate, and gingiva as well as mild generalized swelling in the upper lip. The patient who had no cutaneous findings has a history of VL from 10 years ago. This patient was successfully treated with intravenous sodium stibogluconate.[6] Kharfi et al. reported 5 ML
patients with 4 being female and 1 being male. While only four of the patients had lip mucosa involvement, one patient also had accompanying endonasal mucosa involvement. All the patients responded to intramuscular meglumine antimoniate treatment.[7] The
majority of our patients had cutaneous involvement and no visceral involvement. Only one of the patients had gingival involvement apart from the lip. The dominant type was the lesion type ulcer. The response rate of our patients to intramuscular meglumine antimoniate treatment was 92%.
We are of the opinion that our study will contribute to the literature since it is the largest case report with mucosal involvement in the old world leishmaniasis. When leishmaniasis disease develops especially in people living in endemic areas or travelling to those areas, we should definitely consider the ML disease in cases of treatment‑resistant mucosal (especially lip involvement) lesions. Otherwise, the disease may result in significant morbidity and mortality due to delayed treatments.
Letters to the Editor
Lip leishmaniasis: Clinical characteristics of 621 patients
Table 1: Clinical characteristics of patients with lip leishmaniasis
Frequency Sex
Female 314 (50.56%)
Male 307 (49.44%)
Clinical involvement
Only mucosal involvement 174 (29.1%) Mucosal and cutaneous involvement 447 (71.9%)
Age (years) 15.34±15.26
Lesion duration (weeks) 14.36±9.63 Lesion diameters (millimetres) 14.37±11.83 Number of lesions (quantity) 1.91±1.45 Lesion type Ulcer 425 (68.44%) Nodules 181 (29.15%) Papula 12 (1.93%) Recidivans 3 (0.48%) Treatment
Intramuscular meglumine antimonate
(67 patients=10.7%) 62 (92%)
Intralesional meglumine antimonate
266 International Journal of Critical Illness and Injury Science | Vol. 5 | Issue 4 | Oct-Dec 2015 266
Letters to the Editor
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Yavuz Yeşilova, Mustafa Aksoy1, Hacer Altun Sürücü1,
Ahmet Uluat2, Nurittin Ardic3, Abdullah Yesilova4
Department of Dermatology, Special Lokman Physician Hospital,
4Department of Biostatistics, Yuzuncu Yıl University School of Medicine,
Van, 1Department of Dermatology, Harran University School of Medicine,
2Department of Ear, Nose and Throat, Birecik State Hospital, Sanliurfa,
3Department of Microbiology, Gulhane Military Medical Academy,
Ankara, Turkey
Address for correspondence: Dr. Yavuz Yeşilova,
Department of Dermatology, Special Lokman Physician Hospital, Van 63300, Turkey. E‑mail: [email protected]
REFERENCES
1. Goto H, Lauletta Lindoso JA. Cutaneous and mucocutaneous leishmaniasis. Infect Dis Clin North Am 2012;26:293‑307.
2. Gurel MS, Ulukanligil M, Ozbilge H. Cutaneous leishmaniasis in Sanliurfa:
Epidemiologic and clinical features of the last four years (1997‑2000). Int J Dermatol 2002;41:32‑7.
3. Manual for Surveillance of American Integumentary Leishmaniasis.
Series A. Technical Standards and Manuals. Ministry of Health,
Department of Health Surveillance/Brazil. 2nd ed. 2010. [Updated 1st
reprint].
4. Amato VS, Tuon FF, Siqueira AM, Nicodemo AC, Neto VA. Treatment of
mucosal leishmaniasis in Latin America: Systematic review. Am J Trop Med Hyg 2007;77:266‑74.
5. Ibrahim M, Suliman A, Hashim FA, el Khalil TA, Evans DA, Kharazmi A,
et al. Oronasal leishmaniasis caused by a parasite with an unusual isoenzyme profile. Am J Trop Med Hyg 1997;56:96‑8.
6. Abbas K, Musatafa MA, Abass S, Kheir MM, Mukhtar M, Elamin EM, et al. Mucosal leishmaniasis in a Sudanese patient. Am J Trop Med Hyg 2009;80:935‑8.
7. Kharfi M, Fazaa B, Chaker E, Kamoun MR. Mucosal localization of leishmaniasis in Tunisia: 5 cases. Ann Dermatol Venereol 2003;130 (1 Pt 1):27‑30.
Cite this article as: Yesilova Y, Aksoy M, Surucu HA, Uluat A, Ardic N,
Yesilova A. Lip leishmaniasis: Clinical characteristics of 621 patients. Int J Crit Illn Inj Sci 2015;5:265-6.
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Access this article online Website: www.ijciis.org Quick Response Code: DOI: 10.4103/2229-5151.170849
High voltage electrical shock with multiple
life‑threatening injuries
Dear Editor,
In unprotected form, electricity has the potential to produce devastating injuries with heavy functional and esthetic consequences.[1] The reported incidence of
electrical current related burn injuries ranges from 3 to 17% of all admissions in burn units.[2] Because of widespread
availability and commercial utilization of electrical current, there is increase in electrical current‑related injuries from India.[3] A 26‑year‑old male was brought
to the emergency room with the alleged history of burn injuries due to accidental fall of high voltage electric wire on his right shoulder. He tried to keep away the electric wire with his right hand and felt an electric shock. Following which he fell down on the ground. Since the time of injury, the patient was drowsy but arousable. There was no history of vomiting or seizures. On examination, he had tachycardia (pulse rate 120/min). Respiratory rate was 24 breaths/min, temperature was 99oF, and blood pressure was 110/90 mmHg. Oxygen
saturation was 98% at room air. His chest examination was normal. Cranial nerves were normal. There were no focal neurological deficits. Random blood sugar was
97 mg/dL. Local examination revealed totally charred right upper limb, second‑degree burns were extending up to the shoulder [Figure 1]. Brachial and radial pulses were not palpable. There was amputation of middle finger proximal and distal phalanges, and near total amputation of index finger at distal inter phalangeal joint (DIJ) of right hand [Figure 2a]. There was complete soft tissue loss around proximal phalanges of ring and little fingers of left hand [Figure 2b]. Left ulnar and radial pulses were absent; however, the brachial pulse was present. There were second‑degree burns over the posterior aspect of trunk, head, and neck. There was exit wound over the right iliac region with an anterior abdominal wall defect (8 × 6 cm) and exposed intestinal parts [Figure 3]. Arterial color Doppler showed no flow in right brachial, radial, and ulnar arteries. In left upper limb, there was normal biphasic flow in axillary; and brachial arteries, there was no blood flow in radial and ulnar arteries. In right lower limb there was trickle flow in anterior tibial, posterior tibial, and dorsalis pedis arteries. The patient underwent wound debridement. On laparotomy a segment of ileum, 25 cm from ileocolic
View publication stats View publication stats