ContentslistsavailableatScienceDirect
Pathology
–
Research
and
Practice
j o ur na l h o me p a g e :w w w . e l s e v i e r . c o m / l o c a t e / p r p
Original
article
Metastasis
suppressor
proteins
in
cutaneous
squamous
cell
carcinoma
Onder
Bozdogan
a,
Ibrahim
Vargel
b,
Tarik
Cavusoglu
c,
Ayse
A.
Karabulut
d,
Gurbet
Karahan
e,
Nilufer
Sayar
f,
Pınar
Atasoy
g,
Isik
G.
Yulug
e,∗aAnkaraNumuneEducationandResearchHospital,DepartmentofPathology,Ankara,Turkey
bHacettepeUniversity,MedicalFaculty,DepartmentofPlasticSurgery,ScienceInstitute,DepartmentofBioengineering,Ankara,Turkey cPrivatePractice,PlasticSurgery,Ankara,Turkey
dKırıkkaleUniversity,FacultyofMedicine,DepartmentofDermatology,Kırıkkale,Turkey
eBilkentUniversity,FacultyofScience,DepartmentofMolecularBiologyandGenetics,Ankara,Turkey fIstanbulMedipolUniversity,InternationalSchoolofMedicine,DepartmentofPhysiology,Istanbul,Turkey gKırıkkaleUniversity,FacultyofMedicine,DepartmentofPathology,Kırıkkale,Turkey
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:Received9August2015 Receivedinrevisedform 15November2015 Accepted3December2015 Keywords:
Skin
Squamouscellcarcinoma Metastasissuppressorproteins A-431
HaCaT
a
b
s
t
r
a
c
t
Cutaneoussquamouscellcarcinomas(cSCCs)arecommonhumancarcinomas.Despitehaving metasta-sizingcapacities,theyusuallyshowlessaggressiveprogressioncomparedtosquamouscellcarcinoma (SCC)ofotherorgans.Metastasissuppressorproteins(MSPs)areagroupofproteinsthatcontroland slow-downthemetastaticprocess.Inthisstudy,weestablishedtheimportanceofsevenwell-defined MSPsincludingNDRG1,NM23-H1,RhoGDI2,E-cadherin,CD82/KAI1,MKK4,andAKAP12incSCCs.
ProteinexpressionlevelsoftheselectedMSPsweredetectedin32cSCCs,6insituSCCs,andtwoskincell lines(HaCaT,A-431)byimmunohistochemistry.Theresultswereevaluatedsemi-quantitativelyusing theHSCOREsystem.Inaddition,mRNAexpressionlevelsweredetectedbyqRT-PCRinthecelllines.
TheHSCOREsofNM23-H1weresimilarincSCCsandnormalskintissues,whileRGHOGDI2,E-cadherin andAKAP12weresignificantlydownregulatedincSCCscomparedtonormalskin.ThelevelsofMKK4, NDRG1andCD82werepartiallyconservedincSCCs.InstageISCCs,nuclearstainingofNM23-H1 (NM23-H1nuc)wassignificantlylowerthaninstageII/IIISCCs.OnlynuclearstainingofMKK4(MKK4nuc)showed significantlyhigherscoresininsitucarcinomascomparedtoinvasiveSCCs.
Inconclusion,similartootherhumantumors,wehavedemonstratedcomplexdifferential expres-sionpatternsfortheMSPsinin-situandinvasivecSCCs.ThiscomplexMSPsignaturewarrantsfurther biologicalandexperimentalpathwayresearch.
©2016ElsevierGmbH.Allrightsreserved.
1. Introduction
Non-melanomaskin cancers (NMSC) are themost common humanmalignantneoplasms,andcreatesignificantmedical, eco-nomical,andsocialproblemsforthehealthcareservicesworldwide
[1–3].AlthoughthereareothertypesofNMSC,thistermcommonly referstothetwocommonneoplasmsofcutaneoussquamouscell carcinoma(cSCC)andbasalcellcarcinoma(BCC)[1].BCCsareslow growing,malignant,yetrarelymetastasizingcarcinomas [4].On thecontrary,cSCCsaremoreaggressiveinbehavior,andhave con-siderablyhighermetastaticcapacitiesthanBCCs[4].
Metastasisisaverycomplexandmultistepbiologicalprocess directed byvarious proteinsand pathways[5]. Though various
∗ Correspondingauthorat:BilkentUniversity,FacultyofScience,Departmentof MolecularBiologyandGenetics,TR-06800Ankara,Turkey.
E-mailaddress:[email protected](I.G.Yulug).
proteinssupportmetastasis,agroupofproteinscalledmetastasis suppressorproteins(MSPs)specificallyinhibitorslowmetastasis
[6,7].Asadefinition,pureMSPsshouldonlysuppressmetastasis withoutanyeffectontumorigenicity(e.g.proliferation).However, in the complex environment of a cell, theyusually have other importantpropertiesastumorsuppressoractivities,affecting car-cinogenesis, besidestheseMSP functions[6].cSCCs differfrom internalidenticalorgancancersinthattheyhavelowermetastatic ratesandresultinbetterprognosis[4].Thus,cSCCsareinteresting biologicalmodelsfortheresearchof metastasissuppressors.To establishtheimportanceoftheMSPsinnon-melanomaskin can-cer,weselectedsevenessentialandwell-definedMSPs(NDRG1, NM23-H1,RhoGDI2,E-cadherin,CD82/KAI1,MKK4,andAKAP12) thataffectdifferentstepsofmetastasis.
The main aim of this study was to analyze the expression patternsofthesesevenimportantMSPsthat maycontributeto the inhibition of metastasis pathways in cSCCs, as well as in squamouscelllines.Wealsoestablishedtheassociationbetween
http://dx.doi.org/10.1016/j.prp.2015.12.018 0344-0338/©2016ElsevierGmbH.Allrightsreserved.
theseproteins and important clinicopathological parameters in cSCC.
2. Materialsandmethods 2.1. Studygroups
Atotalof38SCCscomposedof32tissuesamplesofclassical squamouscellcarcinoma(SCC-NOS)oftheskinand6insitu carci-nomatissues,obtainedfrom37patients(26M/11F),wereincluded inthisstudy.AllpatientswereCaucasians,andthedetailed char-acteristicsofthestudygrouparesummarizedinAppendix1.SCCs weregradedbyfour-tieredsystemaswell(Grade1),moderately (Grade2)poorlydifferentiated(Grade3)andanaplasticor undif-ferentiatedtumors(Grade4)[8].DatafortheSCCswerecollected byusingtheCAP(TheCollegeofAmericanPathologists)protocols forsquamouscarcinomaoftheskin(www.cap.org).
2.2. Normalskincontrolgroup
Tennormalskintissues(4M/6F)fromreconstructiveoperations, confirmedasnormalbymicroscopy,wereincludedasthenormal tissuegroup.Thenormalgroupincludedskintissuesfromdifferent localizations;twofromtheface,threefromtheextremities,three fromthebreast,andtwofromtheabdomen.Thiscontrolgroupwas thesameastheoneusedinanotherpreviousstudyofourgroup
[9].Normal,non-lesionalepidermisadjacenttoSCC(N-SCC)was alsointegratedinthisstudy.
2.3. Celllines
Thenormal immortalizedkeratinocyte cellline(HaCaT) and thevulvarsquamous carcinomacellline(A-431) wereusedfor immunohistochemistryandquantitativereversetranscriptase-PCR (qRT-PCR)studies[10,11].
2.4. Immunohistochemistrytechniqueandanalysis
Theimmunostainingprocedurewasperformedbytheclassical labeledstreptavidin-biotinimmuno-enzymaticantigendetection system(UltraVision-ThermoScientific;Waltham,MA,USA)with DABchromogen,intheThermo-ShandonSequenza®manual
stain-ingstation(Waltham,MA,USA).Theprimaryantibodystepwas skippedforthenegativecontrol.Thesourcesofprimaryantibodies andthetechnicaldetailsaredemonstratedinAppendix2.
All immunohistochemically stained slides were evaluated by external and internal controls. Stained slides were semi-quantitatively evaluated using a specific immunohistochemical histologicalscoretechnique,(HSCORE),withminormodifications. Theanalysistechniquewasdescribedindetailintheliteratureand inourpreviousstudy[9,12].
2.5. Cellculture
TheA-431celllinewasculturedinDulbecco’sMinimal Essen-tial Medium (DMEM-low glucose, Hyclone-Thermo Scientific, Waltham,MA,USA), andHaCaTcelllinewasgrownin DMEM-Highglucose(Hyclone),supplementedwith10%fetalbovineserum (FBS)and50mg/mlpenicillin/streptomycin,at37◦C,andin5% car-bondioxide.
2.6. Real-timePCR(qRT-PCR)
500ng of total RNA was reverse-transcribed using oligo-dT primers with the First Strand cDNA Synthesis Kit (Fermentas,
Thermo,Waltham,MA,USA).AllqRT-PCRexperimentswere per-formedusingtheSYBR®Greenreagent(Thermo)inanMX3005P
thermocycler (Strategene®, Agilent, Santa Clara, CA, USA). The
GAPDH(glyceraldehyde-3-phosphatedehydrogenase)andHPRT1 (hypoxanthinephosphoribosyltransferase1)referencegeneswere selectedfornormalization.DatawasanalyzedusingthefreeREST©
2009software(Qiagen,Hilden,Germany)[13].Thesequencesof primersusedareaccesiblefromourpreviouslypublishedpaper
[9].
2.7. Statisticalanalysis
StatisticalanalyseswereperformedusingthePASW®
Statis-tics18software(Chicago,IL,USA).Thedifferencesbetweenthe HSCOREsofthegroupswerefirstanalyzedwiththenon-parametric Kruskal–Wallisone-wayanalysistest;followedbyMann–Whitney Utestwasapplied.p≤0.05wasacceptedassignificant.The “Bon-ferronicorrection”wasusedtoreducetypeIerrors.Thecorrelation betweentheparameterswasanalyzedbySpearman’scorrelation test,wherer≥0.25andp≤0.05wereacceptedassignificant. 2.8. Ethicsstatement
This study was financially supported by the Scientific and Technical Research Council of Turkey (TUBITAK, grant number SBAG-108S184).TheprojectwasapprovedbytheKırıkkale Uni-versityLocalEthicsCommittee(07.04.2008/2008-039).
3. Results
3.1. Immunohistochemicalstainingresults 3.1.1. Tissuestudy
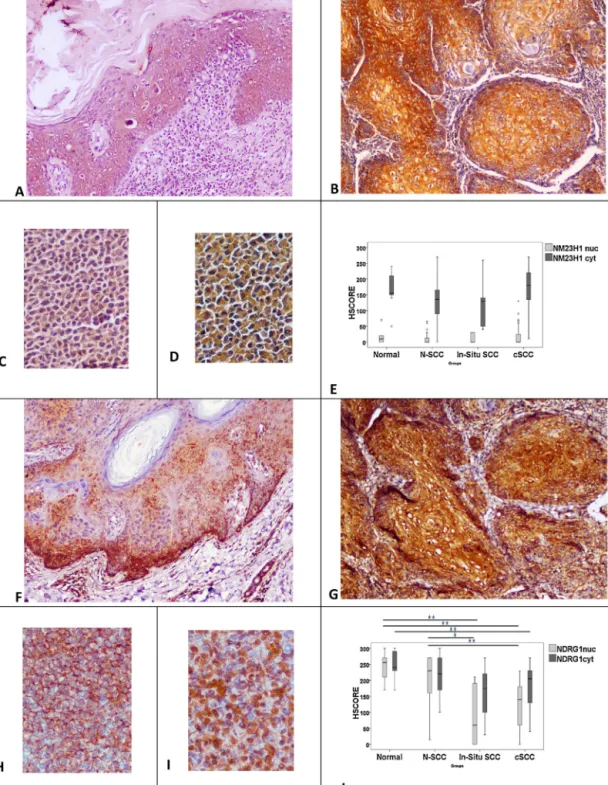
CytoplasmicpositivityofNM23-H1(NM23-H1cyt)andNDRG1
(NDRG1cyt)wasdetectedtobestrongandhomogeneousinallof
theinsituandinvasivecSCCs(Fig.1A,B,F,G).However,nuclear NM23-H1(NM23-H1nuc)expressionwassignificantlyweakerand
wasdetectedinonlytwoofinsituSCCs(IS-SCC,33.3%)and15cSCCs (46.8%) compared tonormal skin.Nuclear positivity of NDRG1 (NDRG1nuc)wasseenin4of6IS-SCCs(66.6%),and29of32cSCCs
(90.6%).
E-cadherin,CD82and AKAP12proteinsshowonlyweakand medium cytoplasmic-membranous staining (Figs. 2A, B, F, G and3A,B).E-cadherinandCD82positivitywasdetectedinallof IS-SCCs,butAKAP12positivitywasobservedin4IS-SCCs(66.6%). E-cadherin,CD82,andAKAP12expressionsweredetectedin84.3%, 68.7%and40.6%ofthecSCCs,respectively.
RHOGDI2showedcytoplasmicpositivityin5 (83.3%)and 30 (93.7%)oftheinsituandinvasivecases,respectively,thoughwith weaktomediumstrengthonly(Fig.3F,G).Nuclearpositivityof RHOGDI2(RHOGDI2nuc)wasweaker,yetmoreheterogeneous,in4
of6IS-SCCs(66.6%)and14of32cSCCs(43.7%).
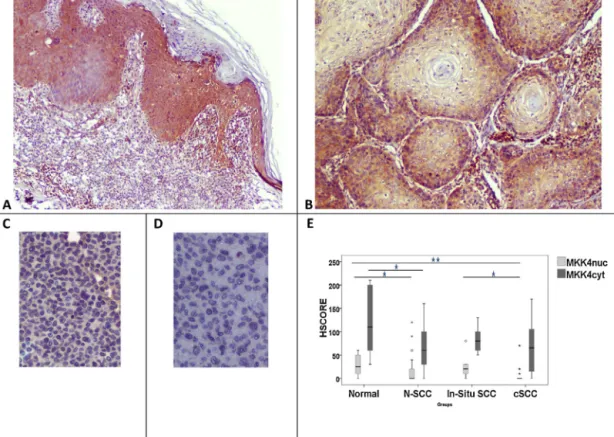
MKK4cytwasdetectedinallofIS-SCCs,andweakMKK4nucwas
detectedin5of6IS-SCCs(83.3%)(Fig.4A).MKK4immunostaining ofcSCCsshowedweaktomediumcytoplasmicpositivityin25cases (78.1%),andweaknuclearpositivityin7cSCCs(21.8%)(Fig.4B). 3.1.2. Celllines
NM23-H1 showed strongcytoplasmic positivity in the both HaCaTandA-431celllines(Fig.1C,D).However,focalnuclear posi-tivitywasstrongerintheHaCaTcellline.Significantpositivitywas observedwithNDRG1antibodyinbothofthecelllines(Fig.1H, I).E-cadherinexpressionwasstrongerintheHaCaTcellline com-paredtoA-431(Fig.2C,D).CD82/KAI1showedmediumlevelsof positivityinbothcelllines(Fig.2H,I).RHOGDI2showedstrongbut heterogeneouspositivityintheHaCaTandA-431celllines(Fig.3H,
Fig.1. NM23-H1expressionininsitu(A)andinvasivecarcinoma(B).Besidesstrongcytoplasmicexpression,weaknuclearexpressionofNM23-H1isoccasionallydetectedin invasivecarcinoma(B).HaCaT(C)andA-431cells(D),bothshowcytoplasmicNM23-H1expression,butstaininginA-431isstronger.Thereisnostatisticaldifferencebetween HSCOREsofthegroups(E).NDRG1expressionininsitucarcinoma(F).Althoughcytoplasmicexpressionisheterogeneous,itisgenerallyhigh.Focalnuclearexpressionis alsodetected.Similarly,cytoplasmicexpressionofNDRG1isverystronginSCCs,butnuclearstainingisdownregulated(G).HaCaTcellsshowcytoplasmicexpression(H) whereasnuclearpositivityisdominantinA-431cellline(I).HSCOREsshowsignificantdownregulationinSCCscomparedtonormalgroups(J).*Showspvalues;*p<0.05; **p<0.01.Originalmagnification:A;B;F;G×100,C;D;H;I×200.
I).AKAP12andMKK4stainingwaseitherveryweakorcompletely lostinbothcelllines(Figs.3C,Dand4C,D).
3.1.3. Comparativestatistics
WhenHSCOREs of normal epidermis(N) were compared to insitucarcinomas,E-cadherin(p=0.016),RHOGDI2nuc(p=0.002),
and NDRG1nuc (p=0.003) stainings showed significantly lower
scoresin insitu carcinomas. However,whencompared to nor-malepidermisadjacenttoSCC(N-SCC),onlyHSCOREsofNDRG1nuc
weresignificantlylowerininsitucarcinomas(p=0.011).
NDRG1nuc/cyt (p=0.001/p=0.007), E-cadherin (p=0.001),
RHOGDI2nuc/cyt (p=0.001/p=0.001), MKK4nuc (p=0.001), and
AKAP12 (p=0.001) expressions were decreased in cSCC com-paredtoN.WhenHSCOREs ofN-SCCwere comparedtocSCCs,
Fig.2.MembranousE-cadherinexpressionininsitu(A)andinvasive(B)carcinoma.ThedownregulationiseasilydetectedinSCC.A-431,squamouscarcinomacellline, showsveryweakpositivity(D)whencomparedtoHaCaT(C).ThedifferencebetweenthenormalskinandinsitucarcinomaisstatisticallysignificantforE-cadherinstaining. Ininsitucarcinomas,theHSCOREsofE-cadherinarelowerthanthenormalepidermis(E).Howeverthereisnostatisticaldifferencebetweeninsitucarcinomaandnormal epidermisadjacenttoneoplasia(N-SCC).InvasiveSCCshowslowerlevelsofE-cadherinexpressionthannormalepidermisandN-SCC.CD82showsmembranocytoplasmic stainingininsitu(F)andinvasiveSCCs(G).InsituSCCsamplesshowmoreheterogenousstainingthaninvasiveSCCs.A-431cellsshowweakerstaining(I)thanHaCaTcells (H).NostatisticaldifferenceisdetectedbetweentheHSCOREsofthegroups(J).*showspvalues.*p<0.05;**p<0.01.Originalmagnification:A×40,B;D;F;G×100,C;H;I ×200.
NDRG1nuc(p=0.001), E-cadherin(p=0.001),and RHOGDI2nuc/cyt
(p=0.001/p=0.001)showedsignificantlylowerscoresincSCCs. NM23-H1nucshowedsignificantlylowerscoresinstageIcSCCs
thanstageII/IIISCCs(p=0.049).MKK4nuc wastheonlyonethat
showedhigher scoresin insitu carcinomas when comparedto invasiveSCCs(p=0.011).
3.1.4. Correlationanalysis
There were limited numbers of positive significant corre-lations (p=0.01 level) between several markers as follows: E-cadherin-RHOGDI2cyt (r=0.452), RHOGDI2cyt-MKK4cyt
(r=0.486), and MKK4nuc-MKK4cyt (r=0.481) in invasive
3.1.5. RealtimePCRresults
NM23-H1 and E-cadherin expression levels did not differ betweenA-431andHaCaTcelllines.Yet,NDRG1expressionwas upregulated (34.4-fold, p=0.001), whereas ARHGDIB (RHOGDI2, −4.7-fold,p=0.001),MKK4(−2.1-fold,p=0.001),CD82/KAI( −2.4-fold,p=0.001)andAKAP12(−9.7-fold,p=0.001)expressionswere downregulatedinA-431cellscomparedtoHaCatcells.
4. Discussion
Inthisstudy,wedemonstrateddifferentexpressionpatternsof MSPsincutaneousSCCs.NM23-H1levelswereconservedininsitu andinvasivecarcinomas,and insquamous carcinomacelllines. AlthoughtheroleofNM23-H1inmetastasissuppressioniswell describedinothercarcinomas,itisnotclearinNMSC[6,7].Similar
Fig.3. AKAP12positivityininsitucarcinoma(A).AKAP12stainingisclearlylostinatypicalcells,andpositivityisseenonlyinmaturebenignkeratinocytes.LowAKAP12 expressioninSCC(B).Veryweak,hardlydetectedpositivityintheHaCaTcellline(C).AKAP12isnegativeintheA-431cellline(D).InvasiveSCCshowsdownregulation comparedtoN(E).RHOGDI2positivityininsitucarcinoma(F).Thereisnosignificantdifferenceininsitucarcinomacomparedtoadjacentepidermis.However,asignificant decreasecanbeseenininvasiveSCCscomparedtothenormalgroup.Inflamatorycellsshowstrongpositivity(G).Strong,mainlycytoplasmicRHOGDI2positivityinHaCaT (H).DownregulatedbutnuclearpositivityinA-431(I).IninsituSCCs,onlyRHOGDI2nucshowssignificantlyreducedlevelscomparedtoN.InvasiveSCCsshowsreduced levelsofRHOGDI2nuc/cytcomparedtobothNandN-SCC(E).*showspvalue.*p≤0.05;**p≤0.01.OriginalMagnification:Fx40,A;B;G×100,D;E;H;I×200.
Fig.4. MKK4expressionininsituandinvasiveSCC(A,B).MKK4cytexpressionisclearlyseenininsitu(A),butshowsweakerstaininginSCCs(B).MKK4showsveryweak
positivityinHaCaT(C),butnopositivityisdetectedinA-431cellline(D).MKK4nucshowssignificantlylowerHSCOREsthannormalepidermisininvasivecSCC(B).However, MKK4cytlevelsaremaintainedincSCCs.Ininsitucarcinomas,MKK4nuc/cytlevelsarealsopositive.MKK4nucshowshigherscoresininsitucarcinomaswithrespectto invasiveSCCs.*showspvalue.*p<0.05;**p<0.01.Originalmagnification:A×40,B×100,C;D×200.
toourfindings,Roetal.[14]andStephensonetal.[15]showed thattheexpressionofNM23-H1waswellconservedincSCCsof theskinandkeratoacanthomas(KA).However,Kanitakisetal.[16]
obtained contradictory results and showed reduced NM23-H1 levelsincSCCs.ThesustainedNM23-H1levelsincSCCsprobably contributetotheirnon-metastasizingfeaturesasobservedinother carcinomas.
Despitebeingdownregulatedincarcinomasamplescompared tonormal tissues,the cytoplasmicNDRG1expression is gener-allymaintainedininsituandinvasiveSCCs.Furthermore,mRNA expression of NDRG1 in the SCC cell line, A-431, was 34-fold increasedcomparedtonormal immortalizedcellline,HaCaT. In accordancewithourfindings,Dangetal.[17]demonstratedthat NDRG1mRNAexpressionincreased5.87-foldinactinickeratoses and7.07-foldincSCCswhencomparedtonormalskin.The impor-tance of NDRG1 expression has been examined in SCCs of the internalorgans, in theliterature.DosSantosetal. [18]showed significantupregulationofNDRG1inoralandoropharyngealSCCs, andconcludedthatNDRG1overexpressionwassignificantly corre-latedwithlong-termsurvival.Similarresultswerealsoreported by Changet al. [19]. Theprognostic importanceof NDRG1was alsodemonstratedinesophagealSCCs,aswellasinotherhuman tumors, suchas tumors of prostate, breast, and colon [20–24]. However, the MSP function of NDRG1 is probably carcinoma-dependent.NDRG1overexpressioninhepatocellularcarcinomahas beenshowntobeanindicatorofpoorprognosis[25].Innonsmall celllungcarcinoma,itsexpressionissignificantlyassociatedwith advancedstagesandweakvascularization[26].
AlthoughHSCOREvaluesofCD82ininsituandinvasive squa-mouscarcinomawerenotdifferentfromnormalskin,only68.7% ofcSCCsshowedCD82positivity,whilelimiteddownregulationof mRNAexpression(2.4-fold)wasdetectedintheA-431cellline.
Similartoourfindings,Okochietal.[27]showedsignificant down-regulation of CD82in basal cellcarcinoma of theskin, despite conserved expression inBowen disease. Thefindings regarding CD82expressioninothertypesofhumanSCCarecontroversial. CD82 downregulation has been demonstrated in oral, cervical, penile,laryngeal,headandneck,andlungSCCs[28–30]. Interest-ingly,when comparedtocSCCs,allofthesecarcinomaspossess greatermetastasispotential.RelativelyconservedlevelsofCD82 maysupportthenon-metastasizingfeaturesofcutaneousSCCs.
Cytoplasmic MKK4 expression was maintained at relatively higherlevelsinSCCsinourstudy.MKK4expressionwas previ-ouslyshowntobereducedduringcancerprogressioninprostate andovarycarcinomas[31,32].Similarresultswereshowninother epithelialcancers,includingendometrialcarcinoma,gastric carci-noma,andpancreaticcancer[33–35].MKK4isgenerallyaccepted tobeametastasissuppressor,buttherearestillconflictingreports intheliterature.Huang etal.foundhigherexpressionofMKK4 inlaryngealSCCsincomparisonwiththeirnormalcounterparts, inadditiontoapositivecorrelationbetweenhigherexpressionof MKK4andincreasedmetastasis[36].Inarecentexperimental arti-cle,Fineganetal.[37]proposedthatMKK4haspro-oncogenicroles inskincarcinoma.OurdatamaysupporttheirhypothesisincSCCs. TheHSCOREsofRHOGDI2weresignificantlydownregulatedin cSCCs.However,RHOGDI2cytoplasmicexpressionwasmaintained ininsitucarcinoma.RHOGDI2expressionhasnotbeenstudiedin skincarcinomaspreviouslyasfarasweareaware.TheMSPfunction ofRHOGDI2wasfirstshowntobeametastasissuppressorgenein bladdercarcinoma[38].Becauseofconflictingreports,the metas-tasissuppressorfunctionofthisproteincouldbeconsideredtobe tissueororgan-dependent.RHOGDI2hasbeenpreviouslyshownto beupregulatedingastricandovariancarcinoma,andprobablyhas adualroleincarcinogenesis[39].
E-cadherinlevelsweresignificantlydownregulated incSCCs. DownregulationofE-cadherinlevelshasbeenwelldemonstrated incSCCbefore[40–42].However,wefoundthatmRNAlevelswere notchangedinthesquamouscellcarcinomacellline.Ithasalso beenreportedthatareducedlevelofE-cadherinexpressionwas moreevidentinacantholyticsubtypesofcSCCs[41].
We demonstrated thatAKAP12 wasdownregulated in cSCC, withlessthanhalfofthecSCCsbeingpositiveforAKAP12staining. SignificantmRNAdownregulationofAKAP12wasalsodetectedin theA-431SCCcellline.ThedecreaseinAKAP12levelsinhuman carcinomashasbeenreportedinvarioustypesofhumancancers
[43].Yet,theexpressionpatternofAKAP12isnotknowninskin car-cinomas.ArecentstudyfocusedonepigeneticregulationofAKAP12 in skincancer and pointed out that thepromoter methylation frequenciesweresignificantlyhigherincarcinomaswithrespect tonormalskintissues.AKAP12promotermethylationfrequencies weredemonstratedtobe89.6%,87.1%and51.2%incSCC,BCCand AK,respectively[44].
Inthisstudy,NM23-H1,NDRG1,RHOGDI2andMKK4antibodies showedbothcytoplasmicandnuclearpositivity.Itiswellknown thatthesubcellularlocalizationoftheproteinsalterstheir func-tions[45].TheimportanceofthisfactisnotclearinMSPs.However, Epstein–Barrvirusnuclearantigen3C(EBNA-3C)promotes Nm23-H1 nuclear localization and modulate Nm23-H1activities [46]. Ismailet al. [47] showedthat nuclearlocalization of NM23-H1 proteininbreastcancerisseenmorefrequentlyinnode-positive metastaticcases.Theyconcludedthatabnormalnuclear accumula-tionoftheproteinmayindicatetumorprogression[47].Similarly, wedemonstratedthat nuclearNM23-H1wassignificantly posi-tivelycorrelatedwithstage.ThenuclearsequestrationofNM23-H1 mayreduceoralter thenormal functionoftheprotein.Besides NM23-H1,theimportanceofsubcellularlocalizationofNDRG1has alsobeenemphasizedintheliterature[48–50].Althoughnuclear positivityofMKK4andRHOGDIhasbeendemonstratedinthe lit-erature,toourknowledge,itsmeaningisnotwellknown[35,51]. 5. Conclusions
Although NM23-H1 expression was maintained at constant levels, RGHOGDI2, E-cadherin, and AKAP12 were significantly downregulatedincSCCs.Furthermore,MKK4,NDRG1andCD82 werealsopartially expressedincSCCs. Similartootherhuman tumors,cSCCsshowadistinctMSPprofile.Data fromthisstudy mightalsorevealpossiblepathwaysamongMSPs,whencombined withthecurrentknowledgeonrelatedpathways.Thisrelationship betweentheseMSPswarrantsfurtherbiologicalandexperimental pathwayresearchtobetterunderstandthemetastaticeventswith regardtoSCCs.
Acknowledgements
This study was financially supported by the Scientific and Technical Research Council of Turkey (TUBITAK, grant number SBAG-108S184).TheprojectwasapprovedbytheKırıkkale Uni-versityLocalEthicsCommittee(07.04.2008/2008-039).
AppendixA. Supplementarydata
Supplementarydataassociatedwiththisarticlecanbefound,in theonlineversion,athttp://dx.doi.org/10.1016/j.prp.2015.12.018. References
[1]A.Lomas,J.Leonardi-Bee,F.Bath-Hextall,Asystematicreviewofworldwide incidenceofnonmelanomaskincancer,Br.J.Dermatol.166(5)(2012) 1069–1080.
[2]V.Madan,J.T.Lear,R.M.Szeimies,Non-melanomaskincancer,Lancet375 (9715)(2010)673–685.
[3]T.S.Housman,etal.,Skincancerisamongthemostcostlyofallcancerstotreat fortheMedicarepopulation,J.Am.Acad.Dermatol.48(3)(2003)425–429. [4]A.S.Weinberg,C.A.Ogle,E.K.Shim,Metastaticcutaneoussquamouscell
carcinoma:anupdate,Dermatol.Surg.33(8)(2007)885–899.
[5]S.Acharyya,L.Matrisian,J.M.D.R.W.,Invasionandmetastasis,in:M.J(Ed.), MolecularBasisofCancer,Elsevier,Philadelphia,2015.
[6]L.A.Shevde,D.R.Welch,Metastasissuppressorpathways–anevolving paradigm,CancerLett.198(1)(2003)1–20.
[7]C.W.Rinker-Schaeffer,etal.,Metastasissuppressorproteins:discovery, molecularmechanisms,andclinicalapplication,Clin.CancerRes.12(13) (2006)3882–3889.
[8]E.Calonje,etal.,Tumorsofthesurfaceepithelium,in:T.B.EduardoCalonje, A.J.Lazar,P.H.McKee(Eds.),PathologyoftheSkinwithClinicalCorrelations, Elsevier,Saunders,Philadeplphia,PA,2012.
[9]O.Bozdogan,etal.,Differentialexpressionpatternsofmetastasissuppressor proteinsinbasalcellcarcinoma,Int.J.Dermatol.54(8)(2015)905–915. [10]P.Boukamp,etal.,Normalkeratinizationinaspontaneouslyimmortalized
aneuploidhumankeratinocytecellline,J.CellBiol.106(3)(1988)761–771. [11]K.Pekkola-Heino,etal.,Radiationresponseofvulvarsquamouscell
carcinoma(UM-SCV-1A,UM-SCV-1B,UM-SCV-2,andA-431)cellsinvitro, CancerRes.49(17)(1989)4876–4878.
[12]D.A.Budwit-Novotny,etal.,Immunohistochemicalanalysesofestrogen receptorinendometrialadenocarcinomausingamonoclonalantibody, CancerRes.46(10)(1986)5419–5425.
[13]M.W.Pfaffl,G.W.Horgan,L.Dempfle,Relativeexpressionsoftwaretool(REST) forgroup-wisecomparisonandstatisticalanalysisofrelativeexpression resultsinreal-timePCR,NucleicAcidsRes.30(9)(2002)pe36.
[14]Y.S.Ro,S.J.Jeong,Expressionofthenucleosidediphosphatekinaseinhuman skincancers:animmunohistochemicalstudy,J.KoreanMed.Sci.10(2) (1995)97–102.
[15]T.J.Stephenson,etal.,‘Anti-metastatic’nm23geneproductexpressionin keratoacanthomaandsquamouscellcarcinoma,Dermatology187(2)(1993) 95–99.
[16]J.Kanitakis,etal.,Expressionofthenm23metastasis-suppressorgene productinskintumors,J.Cutan.Pathol.24(3)(1997)151–156.
[17]C.Dang,etal.,Identificationofdysregulatedgenesincutaneoussquamous cellcarcinoma,Oncol.Rep.16(3)(2006)513–519.
[18]M.DosSantos,etal.,PrognosticsignificanceofNDRG1expressioninoraland oropharyngealsquamouscellcarcinoma,Mol.Biol.Rep.39(12)(2012) 10157–10165.
[19]J.T.Chang,etal.,Identificationofdifferentiallyexpressedgenesinoral squamouscellcarcinoma(OSCC):overexpressionofNPM,CDK1andNDRG1 andunderexpressionofCHES1,Int.J.Cancer114(6)(2005)942–949. [20]T.Ando,etal.,DecreasedexpressionofNDRG1iscorrelatedwithtumor
progressionandpoorprognosisinpatientswithesophagealsquamouscell carcinoma,Dis.Esophagus19(6)(2006)454–458.
[21]S.Bandyopadhyay,etal.,ThetumormetastasissuppressorgeneDrg-1 down-regulatestheexpressionofactivatingtranscriptionfactor3inprostate cancer,CancerRes.66(24)(2006)11983–11990.
[22]S.Bandyopadhyay,etal.,Roleoftheputativetumormetastasissuppressor geneDrg-1inbreastcancerprogression,Oncogene23(33)(2004) 5675–5681.
[23]B.Strzelczyk,etal.,Identificationofhigh-riskstageIIcolorectaltumorsby combinedanalysisoftheNDRG1geneexpressionandthedepthoftumor invasion,Ann.Surg.Oncol.16(5)(2009)1287–1294.
[24]H.Cangul,HypoxiaupregulatestheexpressionoftheNDRG1geneleadingto itsoverexpressioninvarioushumancancers,BMCGenet.5(2004)p27. [25]M.S.Chua,etal.,OverexpressionofNDRG1isanindicatorofpoorprognosisin
hepatocellularcarcinoma,Mod.Pathol.20(1)(2007)76–83.
[26]C.Fan,etal.,IncreasedNDRG1expressionisassociatedwithadvancedT stagesandpoorvascularizationinnon-smallcelllungcancer,Pathol.Oncol. Res.18(3)(2012)549–556.
[27]H.Okochi,etal.,Expressionoftetra-spanstransmembranefamily(CD9,CD37, CD53,CD63,CD81andCD82)innormalandneoplastichumankeratinocytes: anassociationofCD9withalpha3beta1integrin,Br.J.Dermatol.137(6) (1997)856–863.
[28]M.E.Buim,etal.,DownregulationofCD9proteinexpressionisassociated withaggressivebehavioroforalsquamouscellcarcinoma,OralOncol.46(3) (2010)166–171.
[29]Y.Xiong,etal.,ExpressionofmetastasissuppressorgeneKAI1/CD82in cervicalsquamouscellcarcinomaanditsclinicalsignificance,AiZheng24(1) (2005)110–115.
[30]C.Protzel,etal.,Down-regulationofthemetastasissuppressorprotein KAI1/CD82correlateswithoccurrenceofmetastasis,prognosisandpresence ofHPVDNAinhumanpenilesquamouscellcarcinoma,VirchowsArch.452 (4)(2008)369–375.
[31]S.D.Yamada,etal.,Mitogen-activatedproteinkinasekinase4(MKK4)actsas ametastasissuppressorgeneinhumanovariancarcinoma,CancerRes.62 (22)(2002)6717–6723.
[32]H.L.Kim,etal.,Mitogen-activatedproteinkinasekinase4metastasis suppressorgeneexpressionisinverselyrelatedtohistologicalpatternin advancinghumanprostaticcancers,CancerRes.61(7)(2001)2833–2837. [33]M.Ishikawa,etal.,FunctionalandclinicopathologicalanalysisoflossofMKK4
[34]S.C.Cunningham,etal.,MKK4statuspredictssurvivalafterresectionof gastricadenocarcinoma,Arch.Surg.141(11)(2006)1095–1099(discussion 1100).
[35]W.Xin,etal.,MAP2K4/MKK4expressioninpancreaticcancer:genetic validationofimmunohistochemistryandrelationshiptodiseasecourse,Clin. CancerRes.10(24)(2004)8516–8520.
[36]C.Huang,etal.,Overexpressionofmitogen-activatedproteinkinasekinase4 andnuclearfactor-kappaBinlaryngealsquamouscellcarcinoma:apotential indicatorforpoorprognosis,Oncol.Rep.22(1)(2009)89–95.
[37]K.G.Finegan,C.Tournier,Themitogen-activatedproteinkinasekinase4hasa pro-oncogenicroleinskincancer,CancerRes.70(14)(2010)5797–5806. [38]J.J.Gildea,etal.,RhoGDI2isaninvasionandmetastasissuppressorgenein
humancancer,CancerRes.62(22)(2002)6418–6423.
[39]E.M.Griner,D.Theodorescu,ThefacesandfriendsofRhoGDI2,Cancer MetastasisRev.31(3–4)(2012)519–528.
[40]L.C.Fuller,etal.,ExpressionofE-cadherininhumanepidermal
non-melanomacutaneoustumours,Br.J.Dermatol.134(1)(1996)28–32. [41]J.R.Griffin,etal.,Decreasedexpressionofintercellularadhesionmoleculesin
acantholyticsquamouscellcarcinomacomparedwithinvasive
well-differentiatedsquamouscellcarcinomaoftheskin,Am.J.Clin.Pathol. 139(4)(2013)442–447.
[42]A.Lyakhovitsky,etal.,ExpressionofE-cadherinandbeta-cateninin cutaneoussquamouscellcarcinomaanditsprecursors,Am.J.Dermatopathol. 26(5)(2004)372–378.
[43]I.H.Gelman,EmergingrolesforSSeCKS/Gravin/AKAP12inthecontrolofcell proliferation,cancermalignancy,andbarriergenesis,GenesCancer1(11) (2010)1147–1156.
[44]W.Wu,etal.,ExaminationofAKAP12promotermethylationinskincancer usingmethylation-sensitivehigh-resolutionmeltinganalysis,Clin.Exp. Dermatol.36(4)(2011)381–385.
[45]C.-S.Yu,etal.,Predictionofproteinsubcellularlocalization,Proteins:Struct. Funct.Bioinform.64(3)(2006)643–651.
[46]A.Saha,E.S.Robertson,Functionalmodulationofthemetastaticsuppressor Nm23-H1byoncogenicviruses,FEBSLett.585(20)(2011)3174–3184. [47]N.I.Ismail,etal.,Nuclearlocalizationandintensityofstainingofnm23protein
isusefulmarkerforbreastcancerprogression,CancerCellInt.8(2008)p6. [48]P.Lachat,etal.,ExpressionofNDRG1,adifferentiation-relatedgene,in
humantissues,Histochem.CellBiol.118(5)(2002)399–408.
[49]A.Kawahara,etal.,NuclearexpressionofN-mycdownstreamregulatedgene 1/Ca(2+)-associatedprotein43iscloselycorrelatedwithtumorangiogenesis andpoorsurvivalinpatientswithgastriccancer,Exp.Ther.Med.2(3)(2011) 471–479.
[50]Y.Song,etal.,CorrelationofN-mycdownstream-regulatedgene1subcellular localizationandlymphnodemetastasesofcolorectalneoplasms,Biochem. Biophys.Res.Commun.439(2)(2013)241–246.
[51]D.Theodorescu,etal.,ReducedexpressionofmetastasissuppressorRhoGDI2 isassociatedwithdecreasedsurvivalforpatientswithbladdercancer,Clin. CancerRes.10(11)(2004)3800–3806.