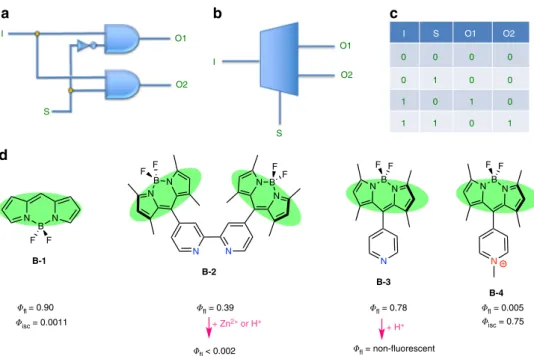
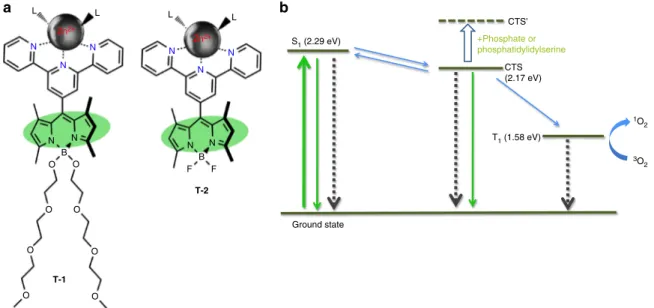
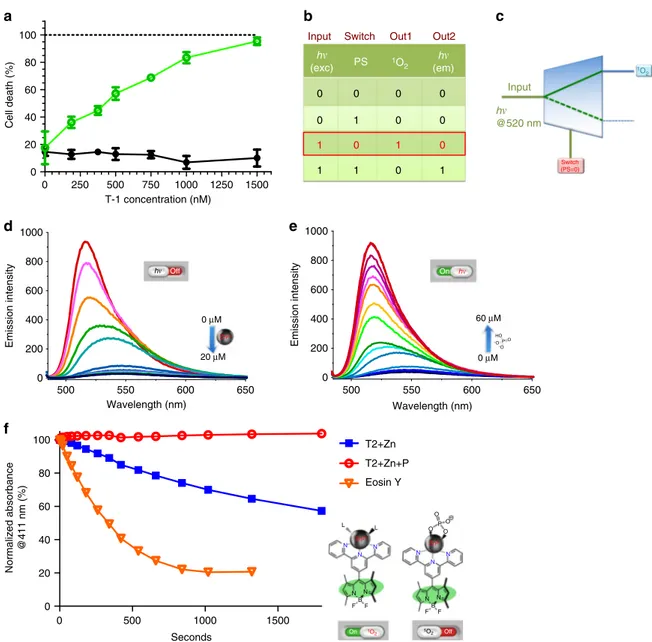
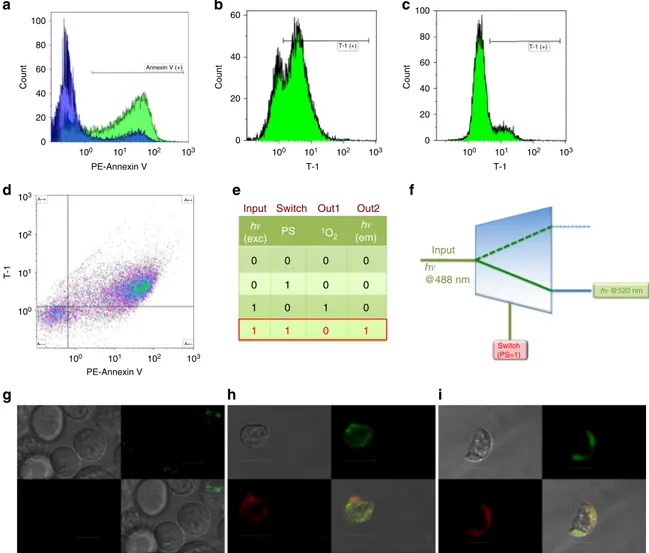
Molecular demultiplexer as a terminator automaton
Tam metin
Şekil
Benzer Belgeler
As an application, we list the deformation families and compute the fundamental groups of all irreducible maximizing simple sextics with a type D singular point.. We attempt to
[r]
In case, there is no other charging station nearby and the primary battery is drained completely the system will automatically switch to the secondary battery and it is being
The energy level-1 includes up to two electrons in spherical orbital named 1s, and energy level-2 holds up to eight electrons, built by two electrons in 2s orbital six electrons in
Specifically, crosslinked network systems of neat epoxy and epoxy-P(St-co-GMA) are modeled to discuss the effect of various molecular interactions as a function of temperature on
For example, in the presence of two target nodes and three jammer nodes, an optimal power allocation strategy according to Scheme 2 can always be obtained by assigning non-zero power
Cofounded by Fred Smith and Henry Rood in September 1 9 1 1 as a nationwide campaign, the Men and Religion Forward Movement (MRFM) emerged out of the cooperation between
However, it was not just the beginning of a new attitude solely towards Turkey but from a larger viewpoint it was, moreover, the beginning of important adjustments in the premises
