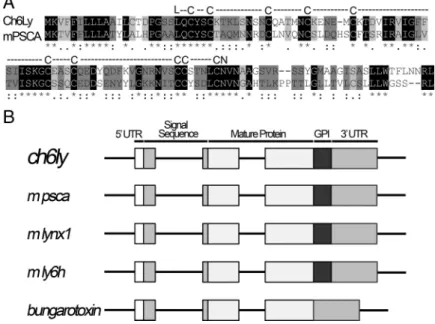
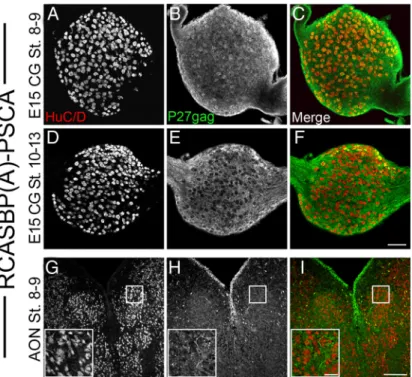
Prostate stem cell antigen is an endogenous lynx1-like prototoxin that antagonizes α7-containing nicotinic receptors and prevents programmed cell death of parasympathetic neurons
Tam metin
Şekil

Benzer Belgeler
Calculated outputs of the model such as activation and inactivation rate functions, activation and inactivation gate variables, ionic channel currents and membrane potential
Changes in the amino acid sequence in the variable region of the heavy and light chain of the Ig molecule. Determines
Oysaki KTÖ ölçeği, MTÖ ölçeğinin eksikliklerine odaklanarak MTÖ ölçeğine alternatif olarak geliştirilmiştir (Yıldırım ve İçeri, 2010:123; Karataş Aracı
Adil fiyat kavramı hakkında ortaya çıkan tartışmaları tarihî süreç içerisinde inceleyen eser, İslam dünyasında yapılan adil fiyat tartışmalarını kapsamı dışında
Officers and the implementation of Field Extension Officers’ duties; 2) knowledge of extension management and the implementation of Field Extension Officers’
Loading the BigData: This is one of the major functionality of the system where we are trying to load a very huge volume data into the HDFS[ Hadoop Distributed File
Immunohistochemistry for S-100 revealed a moderate to strong positive reaction in the cytoplasm of the brown fat tissue cells (Fig. 3-a), but weak positive immune reaction
L- bulanıklaştırılmış esnek komşuluk yapısına L-bulanıklaştırılmış topolojisi tarafından üretilen esnek dönüşüm ve üçlüsüne de L-