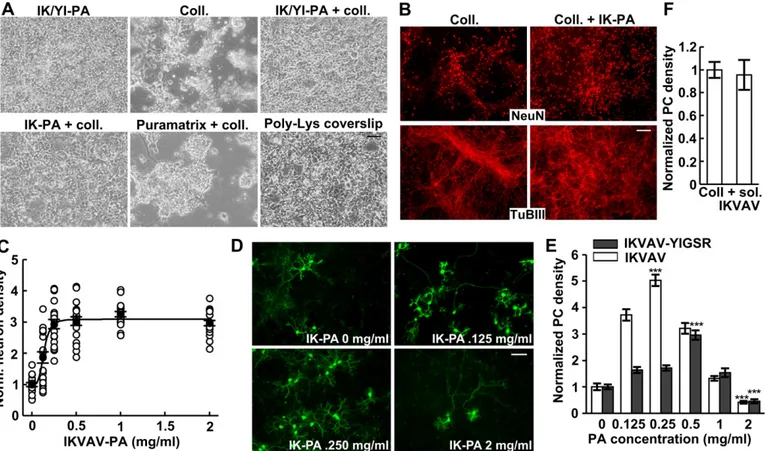
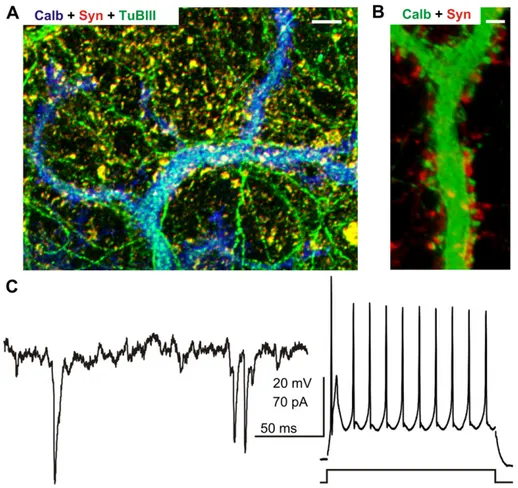
A hybrid nanofiber matrix to control the survival and maturation of brain neurons
Tam metin
Şekil


Benzer Belgeler
The calculation of the velocity field in the flow around cylindrical bodies can be based on the method of expansion of the vector potential of this field.. ( V =
aforementioned skills, if there is a good cooperation within the team and if the participating surgeon has some interests in transcatheter procedures, which should be the case in
The purpose of the present study is to determine whether the domains of disconnection/rejec- tion and impaired autonomy mediate the relationship be- tween childhood
Consequently in the future options put forth in Egypt regarding the Caliphate, both the Scholars Solidarity Delegation and the Alexandrian Committee on the Caliphate
Daha sonra bu kara delikler birleşip, öteki yıldız artıklarıy- la beslenip büyüyerek süper kütleli kara delikler haline geliyorlar.. Bu varsayımın doğruluğunu sına- mak
Sonuç olarak; bas›n-yay›n kurulufllar› ve e¤i- tim kurumlar›na ilave olarak baflta birinci ba- samak sa¤l›k kurulufllar› olmak üzere tüm sa¤l›k
Mostly presynaptic cell releases a chemical signal that diffuses across cleft an binds to a membrane on postsynaptic cell.. Also human can contains electrical synapses where two cells
Hence, this study aims to explore both students and English teachers' conception of the traits and behavior of the good teacher hoping that this will encourage teachers to