Başlık: Detection of Coxiella burnetii from ticks by Polymerase Chain Reaction and Restriction Fragment Length PolymorphismYazar(lar):ALTAY ÇAPIN, Gülay; EMRE, Zişan; CANPOLAT, Seyit; VATANSEVER, Yusuf; DÜZGÜN, Ali Cilt: 60 Sayı: 4 Sayfa: 263-268 DOI: 10
Tam metin
Şekil

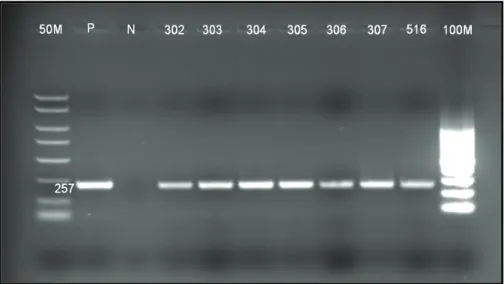
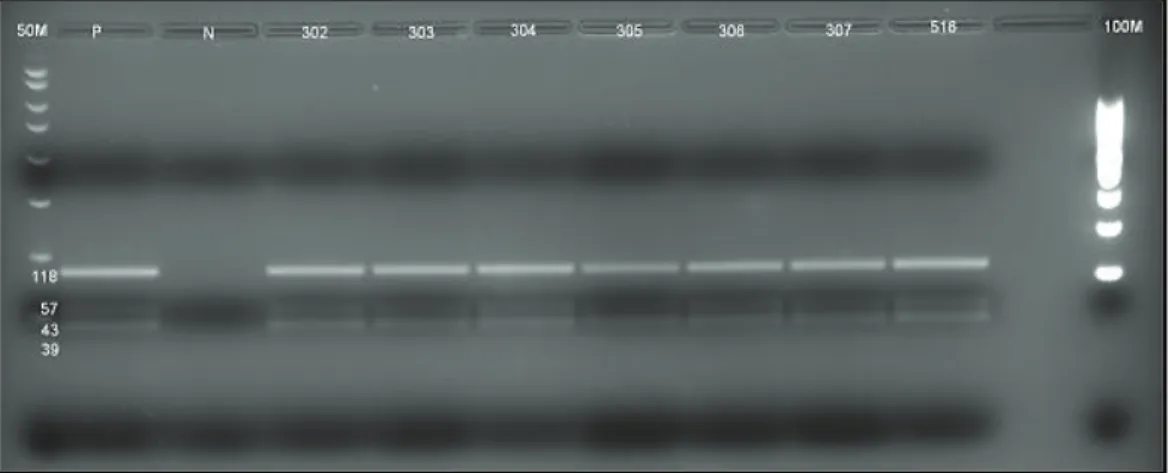
Benzer Belgeler
Çalışmamızda DSM-IV (Ruhsal Bozuklukların Tanısal ve Sayımsal El Kitabı) kriterlerine (19) göre şizofreni tanısı konmuş, 50 (25 kadın ve 25 erkek)
Intracellular DNA replication • DNA replication occurs at 37 ° C • Helper proteins like single strand binding proteins are used in replication • A RNA primer of 12 nucleotide
To observe binding interactions between single-walled carbon nanotubes and major PCR components as DNA template, DNA polymerase enzyme and primers, magnetic beads
The aim of the present study was to investigate whether bacterial infectious agents such as Chlamydophila pneumoniae and Mycoplasma pneumoniae and viral agents such as
While various creators considered the big data eminence issues, our effort varies in that it centers on the issue of recognizing the items that imply a
Bu çalışmada, servikal atipi saptanan sitoloji örneklerinde tek oturumlu ve “nested” konsensus polimeraz zincir reaksiyonu (PCR) yöntemleri uygulanarak HPV
Sonuç olarak, gerek kan gerekse BACTEC kan kültür şişeleri ile hazırlanan simüle örneklerde 5S rDNA genine özgü PCon 1 ve PCon 2 öncüllerinin kullanıldığı PCR yöntemi ile
Çocuklarda ABM olgularının beyin omurilik sıvılarından (BOS) en sık olarak sırasıyla Haemophilus influenzae tip b, Neisseria meningitidis ve Streptococcus pneumoniae 3,4