The efficacy and safety of omalizumab in refractory chronspontaneous urticaria: Real-life experience in Turkey
Tam metin
Şekil
Benzer Belgeler
Araştırmacı tarafından literatür bilgileri incelenerek hazırlanan Histerektomi Öncesi Anket Formu, Histerektomi Sonrası Anket Formu ve Arizona Cinsel Yaşantılar Ölçeği
This case demonstrates that endovascular stent-grafting should be a valid therapeutic alternative in the management of urgent treatment of abdominal aortic aneurysm in
The incidental finding of pulmonary lesion in patients who were referred for cardiac surgery is 0.6 per cent in this series and would stress the impotance of careful
An anteroposterior radiography with the central x-ray beam angled 25 degrees cephalad or an axillary lateral radiograph may be needed to detect
BMT Güvenlik Şurasında 1992 sonrasında yapılan görüşmelerin ne ya- zık ki, doğuran yani Ermenistan’ın Azerbaycan arazilerine saldırısına göz yu- marak onun yalnız
• Increased risk of bleeding: Aspirin irreversibly inhibits COX-1-mediated TXA2 formation, while other NSAIDs reversibly inhibit the production of TXA2.. Because of the decrease in
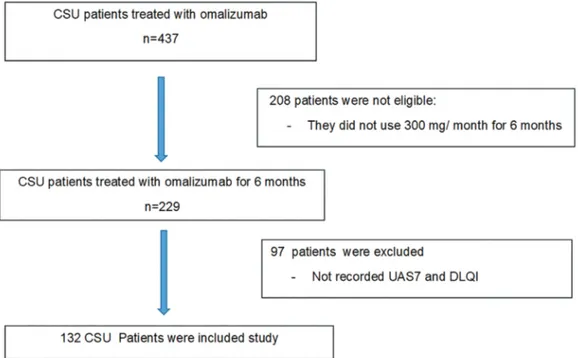
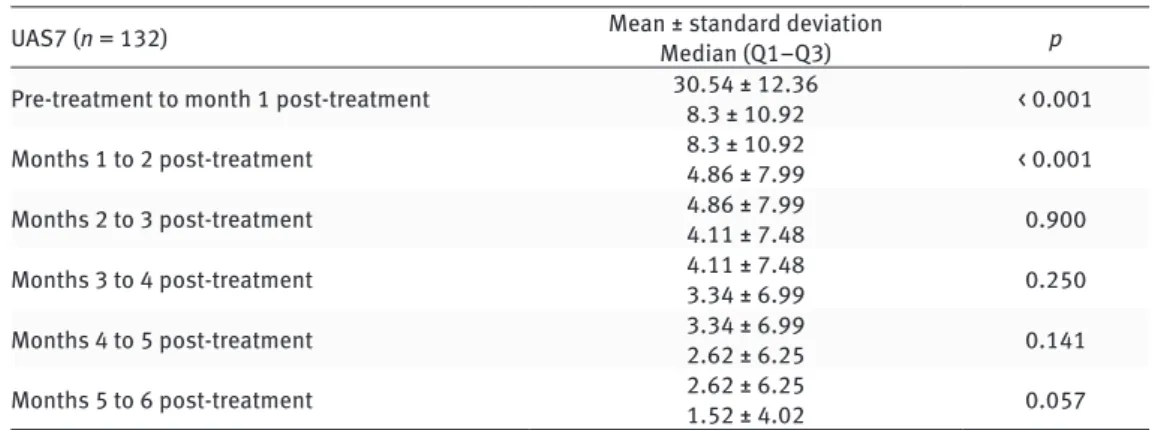
The aim of this study was to evaluate the efficacy and possible side effects of omalizumab, the period from the discontinuation of treatment to the relapse of symptoms, and
As a result, in this study which covers a long period such as 50 months, we observed that omalizumab is rapid acting, effective and safe treatment for chronic