hydrogels Surfactant ion effects and metal on the mechanical properties ofalginate International Journal of Biological Macromolecules
Tam metin
Şekil
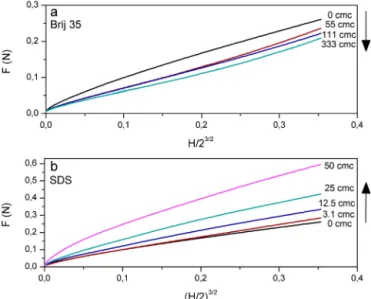
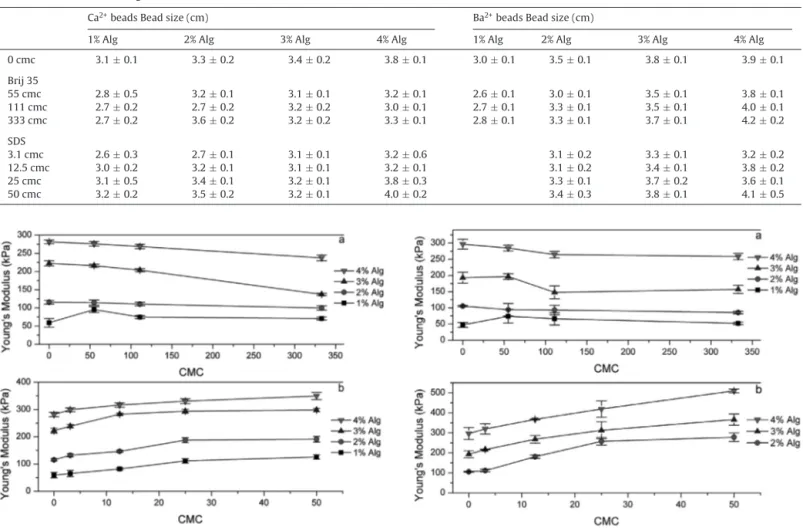
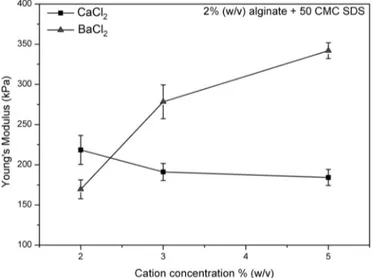
Benzer Belgeler
The disruption of the calcium alginate matrix occurred faster in a phosphate buffer above pH 5.5 because of the chelating action of phosphate ions and at pH 7.4, the affinity
SEM images of (a) pure sodium alginate, (b) pure PVA, (c) 8 mL glutaraldehyde crosslinked SA/PVA hydrogel, (d) 10 mL glutaraldehyde crosslinked SA/PVA hydrogels, (e) 12
The main objective of the current study was to prepare pH- sensitive Ge/SA hydrogels for sustained delivery of an antihistaminic drug (CTZ HCl) to the
to alginate (generally used to prepare beads) in different proportion and found out 1.25:0.75 (alginate:poloxamer) combination in formulation FB4 was the best formulation..
In this study, we compared the slow release coefficients, the encapsulation efficiencies and the initial amount of pyranine in alginate beads crosslinked with different metal (Ca 2+
Desorption of Py in water from the alginate beads cross-linked with calcium ions was studied by using the steady state fluorescence technique.. The fluorescence emission intensity
• In order to increase the effectiveness of inactivated vaccines, these vaccines should be administered in such a way to allow them to stay in the body longer and to stimulate
Although stability characterizes the Turkish-Iranian relationship, especially from 2011 to 2015 the Syrian crisis had revealed the clashing strategic interests of Turkey and Iran