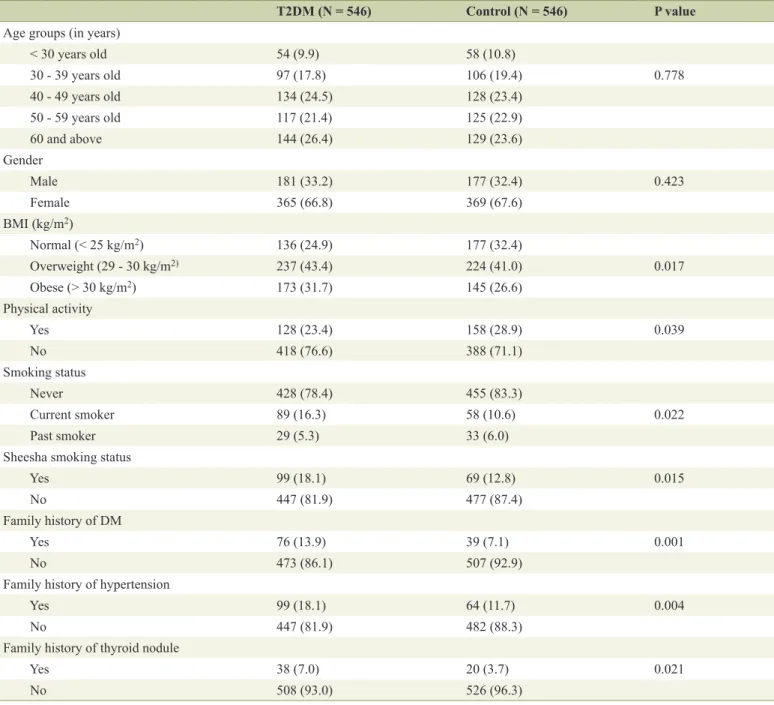
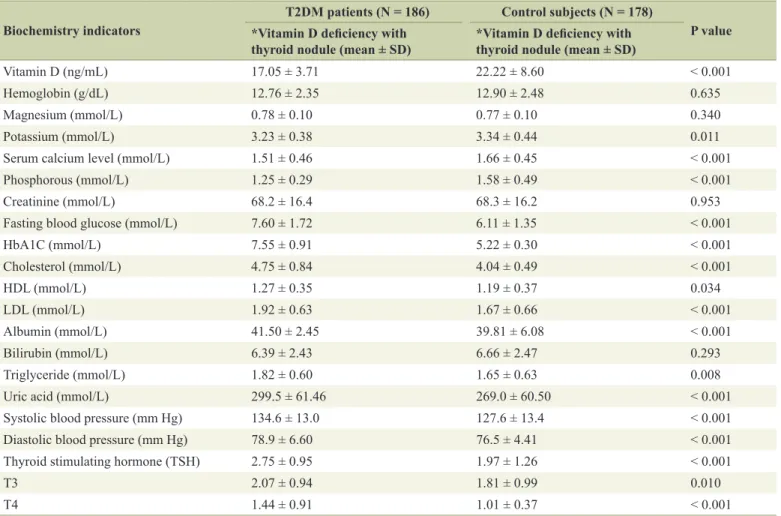
Low vitamin D deficiency associated with thyroid disease among type 2 diabetic mellitus patients
Tam metin
Şekil
Benzer Belgeler
Çalışmamızda HbA1c düzeyinde DEA’sı olan ve olmayan gruplar kıyaslandığında demir eksikliği yerine konduktan sonra HbA1c düzeyinde azalma bulunmuş
Alt ürün molar akış hızı için basamak referans işareti ve gürültüsüz durum için elde edilen benzetim sonuçları Şekil 6-5’te verilmiştir. Şekil 6-5: Alt
Gereç ve Yöntem: Bu çal›flmada, Kocaeli ilinde bulunan 138 Aile sa¤- l›¤› merkezinde çal›flan 420 aile hekimine ve aile sa¤l›¤› elemanlar›na, di¤er aile
Toplumsal cinsiyet rolleri ve değer yargıları, toplumun her alanında etkili ol- duğu gibi kadınların siyasal alana girişinde de önemli rol oynamaktadır. Ka- dınlara seçme
This paper examines the question of the movement or transfer of Cistercian monks between monasteries in England and Wales between 1300 and 1540, with special reference to those
Daha açık bir ifadeyle belirtmek gerekirse hizmet süreç planlama matrisinde, KFG takımı tarafından oluşturulan hiz- met üretim sürecinin her bir adımı ile daha önceki
Rapid diagnosis and effective treatment are required in large vessel vasculitis (LVV) in order to treat symptoms, but more importantly, to reduce the risk of complications such
pediatric and adult RMS, such as (1) the lack of stan- dard management, (2) decreased tolerance to intensive CT protocols because of change in pharmacokinetics and