Summary
The distribution and location of endocrine cells were determined in the dorsal, ventral, and splenic lobes of sparrowhawk pancreas using immunohistochemical methods. In this study, pancreatic tissues were stained with anti-insulin, anti-glucagon, anti-somatostatin, anti-gastrin, and anti-serotonin primary antibodies using the streptavidin-biotin-peroxidase method. The results showed that the numbers of glucagon- (alpha cell), insulin- (beta cell), and somatostatin- (delta cell) releasing cells were high and located in the splenic lobe of the pancreas. These endocrine cells were grouped into alpha, beta, and mixed islets. Alpha islets were mainly composed of alpha and delta cells and also occasionally beta cells. Beta islets contained numerous beta cells and a few delta and alpha cells. Furthermore, in the exocrine tissue were showed as only one cell or 2-3 gastrin immunopositive cell groups, whereas serotonin immunopositive cells were not found anywhere in the exocrine and endocrine pancreas. In conclusion, the endocrine islet types, endocrine cell localizations and lobe numbers of pancreas in sparrowhawks are similar to predator bird species, but are determined different to domestic fowls.
Keywords: Insulin, Glucagon, Somatostatin, Gastrin, Pancreas, Sparrowhawk
Atmaca (Accipiter nisus) Pankreas Dokusu Endokrin
Hücrelerin Dağılımı ve Lokalizasyonu
Özet
Bu çalışmada, atmaca pankreasının dorsal, ventral ve splenik loblarında bulunan endokrin hücrelerin dağılımı immun-histokimysal metotlarla belirlenmiştir. Pankreas dokusu anti-glukagon, anti-insulin, anti-gastrin ve anti-somatostatin primer antikorları kullanılarak streptavidin-biotin peroxidase metodu ile boyandı. Çalışmada, glukagon (alfa hücresi), insülin (beta hücresi) ve somatostatin (delta hücresi) salgılayan hücrelerin splenik lobda sayısının en fazla olduğu saptandı. Atmalarda pankreasın endokrin bölümlerinin alfa, beta ve miks adacıklar halinde gruplaştıkları saptandı. Beta adacıklarının çok sayıda beta hücresi, az sayıda delta ve alfa hücrelerini içerdiği belirlendi. Alfa adacıkları, genellikle alfa ve delta hücrelerinden nadiren de beta hücrelerinden oluşuyordu. Ayrıca, gastrin pozitif hücreler, sadece ekzokrin pankreasta 1 ya da 2-3 adet hücre grubu halinde bulunurken, seratonin pozitif hücrelere ise ekzokrin ve endokrin pankreasın herhangi bir bölümünde rastlanmadı. Sonuç olarak, atmaca pankreasının lop sayısı, endokrin adacık tipleri ve endokrin hücre lokalizasyonu açısından etçil kuşlarla benzerliği, evcil kuş türleriyle de farklılıkları belirlenmiştir.
Anahtar sözcükler: İnsulin, Glukagon, Somatostatin, Gastrin, Pankreas, Atmaca
Distribution and Location of Endocrine Cells in the Pancreas
of the Sparrowhawk, Accipiter nisus
Adem KARA
1
Deniz TEKİNER
2Nejdet ŞİMŞEK
3Hülya BALKAYA
4Zekeriya ÖZÜDOĞRU
41
2
3 4
Atatürk University, Faculty of Veterinary Medicine, Department of Histology and Embryology, TR-25240 Erzurum - TURKEY
Kırıkkale University, Faculty of Veterinary Medicine, Department of Histology and Embryology, TR-71100 Kırıkkale - TURKEY
Balıkesir University, Faculty of Medicine, Department of Histology and Embryology, TR-10100 Balıkesir - TURKEY Atatürk University, Faculty of Veterinary Medicine, Department of Anatomy, TR-25240 Erzurum - TURKEY
Makale Kodu (Article Code): KVFD-2013-10096
The pancreas consists of exocrine and endocrine
(Langerhans islets) compartments, which both have different functions. Mammalians pancreases are comprised of head, body and tail regions [1,2], whereas pancreas of
INTRODUCTION
İletişim (Correspondence)
+90 442 2315536avian species consisted of three [3,4] or four lobes: dorsal ventral, third and splenic [4,5]. Also, the islets of Langerhans of avian species are different from those of mammals [6-8]. Avian islets of Langerhans are divided into alpha, beta, and mixed islets according to their cellular composition [6,7]. Each islet of Langerhans in mammalian pancreas consists of a few to several thousand cells [9,10], which are alpha (A, A2, α), beta (B, β), delta (D, δ), and pancreatic polypeptides (PP, F). Some authors have reported that the islets of Langerhans may also contain substance P, neuropeptide Y, serotonin, cholecystokinin-8, galanin [6], gastrin, and ghrelin-immunopositive cells in the some avian species [1]. In the mammalians, beta cells are located generally in the central of islets, and alpha cells located in the periphery of islets in humans, rats, and mice [1], however these cells are opposite position in the pancreatic islets of horses [2], monkey and kangaroo [11]. In the avian species, endocrine parts of pancreas are generally consisted of large diameter alpha islets, higher number of beta islets and a few or have not mixed islets [1,7,11]. Alpha islets consist of numerous alpha and delta cells, whereas beta islets are consist of numerous beta cells, mildly delta cells, rarely a few alpha cells [5,7].
Researchers have studied the anatomical, histological, and histopathological structures of the pancreas. The distributions of the different types of cells in the pancreas of avian species have been demonstrated by immuno-histochemical methods in mynah [12], chickens[13], falcons [7], ducks [3], geese [14], and young Japanese quails [5]. To our knowledge, there is no study showing the histological distribution of endocrine cells in the pancreas of the sparrowhawk (Accipiter nisus). The aim of this study was to determine the distribution of glucagon-, insulin-, somatostatin-, gastrin-, and serotonin-releasing cells in sparrowhawk pancreas by using immunohistochemical-staining techniques.
MATERIAL and METHODS
Nine sparrowhawks with injuries including shotgun wounds, broken legs, and wings or poisoning were recieved from the animal hospital at Atatürk University, Faculty of Veterinary Medicine, Erzurum, Turkey between 2011 and 2013. Their weights ranged from 150 g to 250 g. Sparrowhawks that could not recover from their injuries were euthanized using ether anesthesia, and pancreatic tissue was taken from the dorsal, ventral, and splenic lobe edges of the pancreas. The tissues were fixed in 10% neutral buffered formalin. The fixed materials were dehydrated in a graded series of ethanol and embedded in paraffin wax. Paraffin wax-embedded pancreases were sectioned in series at a thickness of 5-7 µm.
For examination of the histological structure of the tissue, the sections were stained with Crossman modified Mallory’s triple stain [15]. Pancreatic endocrine cells were
also detected by immunohistochemistry using the streptavidin-biotin-peroxidase method. The sections were deparaffinized in xylene and dehydrated in descending alcohols, and then antigen retrieval was performed by heating the slides in ethylene diamine tetra acetic acid (EDTA) buffer (pH:8.0). Endogenous peroxidase activity was blocked with 3% H2O2. Normal bovine serum was used
to block nonspecific binding sites of antibodies, and then sections were incubated with primary antibodies (glucagon (Leica, 1/50 dilution), insulin (Cell Marque), somatostatin (Cell Marque), gastrin (Leica, 1/50 dilution), and serotonin (Dako, 1/50 dilution)) for one hour. Following this, they were incubated with biotinylated secondary antibody and then streptavidin horseradish peroxidase (Dako, Universal LSAB Kit, K0690) for 30 min each. To demonstrate the reactions, 3,3’-diaminobenzidine tetrahydrochloride (DAB, Sigma) was used. Nuclei were stained with Harris’s haema-toxylin, dehydrated through an ethanol series, and then cleared in xylene before being mounted using Entellan (Merck, German).
Immunohistochemical Evaluation Procedure
The binding of antibodies was evaluated using a high-power Nikon i50 light microscope (Nikon, Tokyo, Japan). The sections of all lobes were evaluated for the location of islets and the distribution of pancreatic endocrine cell types. For each animal, ten serial-sectioned pancreas tissue slides were analyzed for endocrine cell types. Evaluations and scoring of endocrine cell distribution or localization were insulin-positive, glucagon positive, somatostatin-positive, gastrin-positive, and serotonin-positive. The scores were derived semi-quantitatively using light microscopy on the preparations from each animal, and were reported as follows: none = −: not detected, +: rare, ++: a few, +++: moderate, ++++: numerous.
RESULTS
Sparrowhawk pancreas, which consisted of dorsal, ventral, and splenic lobes, was located in a horizontal position in the abdomen. Endocrine islets of different sizes and shapes were dispersed throughout the pancreatic tissue. In the immunohistochemical examination, glucagon-
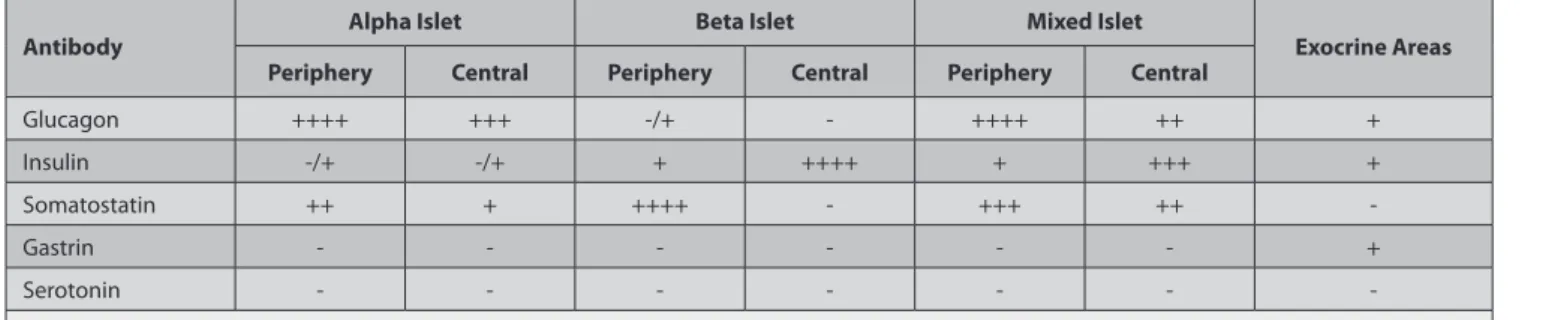
(Fig. 1), insulin- (Fig. 2), and somatostatin - (Fig. 3) immuno-positive endocrine cells were detected in the pancreatic islets. Also, glucagon- (Fig. 1), insulin- (Fig. 2), and gastrin- (Fig. 4) immunopositive cells were found within the acini as both single and ductular areas, but serotonin-immunopositive cells were not detected in either the endocrine pancreas or the exocrine pancreas. The location and distribution of endocrine cells in the pancreas are presented in Table 1.
In the sparrowhawk pancreas, according to the distribution of endocrine cells, there were three types of islets: alpha, beta, and mixed islets. These islets were numerous in the splenic lobe compared to the other lobes.
Alpha islets were found as generally large cell clusters and occasionally small islets, which principally contained alpha cells (Fig. 1), a few delta cells, and occasionally beta cells. In addition, the somatostatin-immunopositive cells were observed in both the peripheral and central regions of the
alpha islets (Fig. 3). Beta islets were generally oval-shaped small endocrine islets. Beta islets were more numerous than alpha islets in all lobes of the sparrowhawk pancreas. Beta islets were comprised mainly of beta cells (Fig. 2)
with a few delta cells located in the periphery of the islets. Fig 1. The distribution of the glucagon
immunopositive cells within the mixed (a) and alpha (b) islets of pancreas; arrows: Some of glucagon immunopositive cells were located in the exocrine region of pancreas. Streptavidin-biotin peroxidase method
Şekil 1. Pankreatic miks (a) ve alfa (b)
ada-lardaki glucagon immun-pozitif hücrelerin adacıklardaki dağılımı, oklar; pankreas ekzokrin bölümünde bazı immunopozitif glukagon hücrelerin dağılımı, Streptavidin-biotin peroxidase metodu
Fig 2. The distribution of the insulin
immunopositive cells within the mixed
(a) and beta (b) islet, and exocrine
pancreas (c), arrows; single located insulin immunopositive cells in the exocrine region of pancreas, Streptavidin-biotin peroxidase method
Şekil 2. Pankreatik miks (a) ve beta (b) adaları ve ekzokrin alan (c) immun-
pozitif İnsülin immunopozitif hücrelerin dağılımı, oklar; ekzokrin pankreasda tek yer-leşimli insulin pozitif hücreler, Streptavidin-biotin peroxidase metot
Fig 3. The distribution of the somatostatin
immunopositive cells within the alpha (a), beta (b) and mixed (c) islets in pancreas, Streptavidin-biotin peroxidase method
Şekil 3. Pankreatik alfa (a), beta (b) ve
miks (c) adalardaki somatostatin immun-pozitif hücrelerin adacıklardaki dağılımı, Streptavidin-biotin peroxidase metot
Mixed islets generally consisted of central-positioned beta cells and peripheral-located alpha cells, or they were occasionally composed of side-by-side alpha and beta islets (Fig. 1 and Fig. 2).
DISCUSSION
This study reveals, for the first time, the existence, location, and distribution of insulin-, glucagon-, somato-statin-, and gastrin-immunopositive endocrine cells in sparrowhawk pancreas using immunohistochemistry methods. The number of islets may vary greatly in size, with large islets being replaced near the arterioles and smaller islets being replaced in the deeper pancreatic parenchyma. Endocrine islets show different distributions, locations, and characteristics among avian species [1]. A high number of alpha-, beta-, and somatostatin-releasing cells, and a small number of gastrin-, serotonin-, substance P-, neuropeptide Y-, and cholecystokinin-8-releasing cells are located in the pancreatic endocrine islets of some avian species [3,5,6,12-14,16]. Therefore, the present study aimed to investigate the distribution and location of endocrine cells in the pancreas of the sparrowhawk.
The Endocrine region of the pancreas in avian species generally consists of large-diameter alpha islets, a higher number of beta islets [6,17], and a few or no mixed islets [5,14]. In many studies, the number of endocrine islets in both avian and mammalian species is higher in the splenic lobe (tail region) than the other lobes of the pancreas [14,18]. According to Rawdon and Larsson [4], small beta islets were found in the dorsal and ventral lobes of the pancreas; moreover, Gulmez et al.[14] and Simsek et al.[5] found
predominantly alpha and beta islets in the splenic lobes of goose and quail pancreas, respectively. In the present study, in sparrowhawk pancreas, alpha, beta, and mixed islets were found in higher numbers in the splenic lobes than in the other lobes.
Glucagon
Pancreatic alpha cells secrete glucagon, which regulates glucose levels in the blood [19]. In this study, glucagon immunopositive cells were located numerously in the throughout of the alpha islets and a few in the periphery of the beta islets and rarely in the exocrine parenchyma. According to Tarakci et al.[8], the alpha cells of ostrich are located in the periphery of the islets. Our findings Fig 4. The distribution of the gastrin
immunopositive cells within the exocrine region of the sparrowhawk pancreas; a,
b, and c; gastrin immunopositive cells are
generally located among the acini as a single in the exocrine region of pancreas, Streptavidin-biotin peroxidase method
Şekil 4. Atmaca pakreası ekzokrin alanında
gastrin immun pozitif hücrelerin dağılımı,
a, b, ve c; ekzokrin pankreas asinuslarına
genellikle tek yerleşimli gastrin immun-pozitif hücreler, Streptavidin-biotin per-oxidase metot
Table 1. Distribution and localization of endocrine cells in pancreas of the sparrowhawk Tablo 1. Atmaca pankreası endoktrin hücrelerinin dağılımı ve lokalizasyonu
Antibody Alpha Islet Beta Islet Mixed Islet Exocrine Areas
Periphery Central Periphery Central Periphery Central
Glucagon ++++ +++ -/+ - ++++ ++ + Insulin -/+ -/+ + ++++ + +++ + Somatostatin ++ + ++++ - +++ ++ -Gastrin - - - + Serotonin - - - -Relative frequencies; −: not detected, +: rare, ++: a few, +++: moderate, ++++: numerous
revealed predominantly in the throughout of the alpha islets in the sparrowhawk pancreas, which is in accordance with previous studies [2,5,6,14]. Also, this study, single or 2-3 alpha cell clusters were observed in the pancreatic parenchyma. However, some authors are demonstrated that alpha cells found only in the endocrine islets of the falcon [7], goose [14] and quail [5]. These differences in locality might be related to paracrine interactions, which may affect the cell types in the different reproductive periods. Paracrine interactions have been proposed as another way of signaling to achieve glucose homeostasis [20]. The different locations of alpha cells in the sparrowhawk pancreas may be related to this mechanism.
Insulin
Each endocrine cell produces only one specific peptide hormone. Beta cells are responsible for the secretion and storage of insulin in response to decreased plasma glucose concentrations. Insulin regulates carbohydrate metabolism and has anti-apoptotic effects on pancreatic cells [21]. In common mammalian species, the beta cells are located in the center of the endocrine islets [1,11]. Conversely, in goose [14], falcons [7], quails [5], chickens [23], and ducks [1], beta cells are mainly located in the beta islets and are rarely observed in the alpha islets. In this study, insulin immunopositive cells were generally located in the beta and mixed islets, and similar to falcon’s [7], single or 2-3 clustered beta cells were located in the alpha islets and exocrine pancreas.
Somatostatin
Somatostatin is a peptide hormone, which is found in neurons, the pancreas, the gut, and some other tissues. There are two forms of somatostatin in the body: SOM-14 and SOM-28, which inhibit both glucagon and insulin secretion [25]. In addition, they inhibit endocrine and exocrine secretions of the pancreas and have an effect on neurotransmission, the gastrointestinal system and biliary motility, intestinal absorption, and cell proliferation [16,26]. Previous studies have showed the existence of delta cells in the pancreases of geese [14], quails [5] and long-legged buzzards [6]. In these studies, somatostatin-releasing cells were found more frequently in alpha islets than in beta islets. Moreover, delta cells were not observed in the exocrine part of the pancreas. Similarly, Tarakci et al.[8] did not observe delta cells in the exocrine pancreas of the ostrich. Contrary to these results, delta cells are also observed in exocrine pancreas of quail and chicken [5,27].
Gastrin
Endocrine gastrin exists in part of the digestive tract, and gastrin secretions are affected by the regulation of stomach motility in the digestive system [28]. In some studies, gastrin stimulated pancreatic growth and differentiation by stimulation of epithelial growth factor (EGF) [29]. Gastrin
or vasoactive intestinal peptide (VIP), is produced in some pancreatic endocrine neoplasms, although these are not found in normal mammalian endocrine islets. However, gastrin or the homologue of cholecystokinin is transiently expressed in the developing pancreas islets, and the highest gastrin expression is produced by the mammalian fetus pancreatic islets during development [30]. In the present study, immunopositive gastrin cells were found in the exocrine region. To date, no avian studies have detected anti-gastrin, and so the differences between avian and mammalian species are considered here. According to the literature, in developing mammalians, due to the effect of EGF, some ductular cells differentiated to gastrin-producing cells [30]. However, these gastrin secretions did not continue into adulthood, whereas the findings of this study showed that these gastrin-immunopositive cells are found in the adult sparrowhawk.
Serotonin
The existence of serotonin-releasing cells in the pancreas of mammals has been reported [31]. In this study, no serotonin immunopositive cells were found in either the exocrine or the endocrine regions of the sparrowhawk pancreas. On the contrary, some studies reported the existence of serotonin immunoreactive cells in the pancreases of ducks [3] and chickens [32]. Many immuno- histochemical studies have showed the similar morphological characteristics between different species, which may reflect the metabolic characteristics of some avian species.
In conclusion, the examination of the endocrine region of the sparrowhawk pancreas has showed that some differences exist among avian species. The endocrine region of the sparrowhawk pancreas consists of alpha, beta, and mixed islets. Although the insulin- and glucagon immunopositive cells were located in the endocrine and exocrine regions, the somatostatin-releasing cells were found as clusters and/or single cells in the endocrine region. And also, the gastrin immunopositive cells were detected as single cells in the exocrine pancreas, whereas serotonin immunopositive cells were not found in the all of the sparrowhawk pancreas.
REFERENCES
1. Kim A, Miller K, Jo J, Kilimnik G, Wojcik P, Hara M: Islet architecture:
A comparative study. Islets, 1, 129-136, 2009.
2. Furuoka H, Ito H, Hamada M, Suwa T, Satoh H, Itakura C:
Immuno-cytochemical component of endocrine cells in pancreatic islets of horses. Jpn J Vet Sci, 51, 35-43, 1989.
3. Lucini C, Castaldo L, Lai O: An immunohistochemical study of the
endocrine pancreas of ducks. Eur J Histochem, 40, 45-52, 1996.
4. Rawdon BB, Larsson LI: Development of hormonal peptides and
processing enzymes in the embryonic avian pancreas with special reference to colocalisation. Histochem Cell Biol, 114, 105-112, 2000.
5. Simsek N, Ozudogru Z, Alabay B: Immunohistochemical studies on
the splenic lobe of the pancreas in young Japanese quails (Coturnix c. japonica). Deut Tierarztl Woch, 115, 189-193, 2008.
6. Bayrakdar A, Yaman M, Atalar O, Gencer Tarakci B, Ceribasi S:
Distribution of neuropeptides in endocrine and exocrine pancreas of long-legged buzzard (Buteo rufinus): An immunohistochemical study. Regul Pept, 166, 121-127, 2011.
7. Simsek N, Bayraktaroglu AG, Altunay H: Localization of insulin
immunpositive cells and histochemical structure of the pancreas in falcons (Falco anaumanni). Ankara Univ Vet Fak Derg, 56, 241-247, 2009.
8. Tarakci BG, Yaman M, Bayrakdar A: Immunohistochemical study on
the endocrine cells in the pancreas of the ostrich (Struthio camelus). J Anim Vet Adv, 6, 693-696, 2007.
9. Iki K, Pour PM: Distribution of pancreatic endocrine cells including
IAPP-expressing cells in non-diabetic and type 2 diabetic cases. J Histochem Cytochem, 55, 111-118, 2007.
10. Ponery A, Adeghate E: Distribution of NPY and SP and their effects
on glucagon secretion from the in vitro normal and diabetic pancreatic tissues. Peptides, 21, 1503-1509, 2000.
11. Steiner DJ, Kim A, Miller K: Pancreatic islet plasticity: Interspecies
comparison of islet architecture and composition. Islets, 2, 135-145, 2010.
12. Saadatfar Z, Asadian M: Anatomy of pancreas in mynah (Acridotheres
tristis). J Appl Anim Res, 36, 191-193, 2009.
13. Cooper KM, Kennedy S, McConnell S, Kennedy DG, Frigg M: An
immunohistochemical study of the distribution of biotin in tissues of pigs and chickens. Res Vet Sci, 63, 219-225, 1997.
14. Gulmez N, Kocamis H, Aslan S, Nazli M: Immunohistochemical
distribution of cells containing insulin, glucagon and somatostatin in the goose (Anser anser) pancreas. Turk J Vet Anim Sci, 28, 403-407, 2004.
15. Bancroft JD CH: Manual of Histological Techniques. pp. 42-45, 71-73,
164-165, Churchill Livingstone Medical Division Longman Group Limited, UK, 1984.
16. Dubois MP: Immunoreactive somatostatin is present in discrete cells
of the endocrine pancreas. PNAS, 72, 1340, 1975.
17. Ku SK LJ, Lee HS: An immunohistochemical study of the insulin-,
glucagon- and somatostatinimmunoreactive cells in the developing pancreas of the chicken embryo. Tissue Cell, 32, 58-65, 2000.
18. Yukawa MTT, Watanabe T, Kitamura S: Proportions of various
endocrine cells in the pancreatic islets of wood mice (Apodemus speciosus). Anat Histol Embryol, 28, 13-16, 1999.
19. Sherwood LM, Parris EE, Unger RH: Glucagon physiology and
pathophysiology. N Engl J Med, 285, 443-449, 1971.
20. Kisanuki K, Kishikawa H, Araki E, Shirotani T, Uehara M, Isami S, Ura S, Jinnouchi H, Miyamura N, Shichiri M: Expression of insulin
receptor on clonal pancreatic alpha cells and its possible role for
insulin-stimulated negative regulation of glucagon secretion. Diabetologia, 38, 422-429, 1995.
21. Chen S, Cheng AC, Wang MS, Zhu DK, Jia RY, Luo QH, Cui HM, Zhou Y, Wang Y, Xu ZW, Chen ZL, Chen XY, Wang XY: Histo-
pathology, immunohistochemistry, in situ apoptosis, and ultrastructure characterization of the digestive and lymphoid organs of new type gosling viral enteritis virus experimentally infected gosling. Poult Sci, 89, 668–680, 2010.
22. Kocamis H, Sari EK, Nazli M, Gulmez N, Aslan S, Deprem T:
Immunohistochemical distribution of insulin-, glucagon-, and somatostatin-containing cells in the pancreas of the rat (Wistar albino). Kafkas Univ Vet Fak Derg, 15, 611-614, 2009.
23. Bagnell CA BN, McMurtry JP: Immunohistochemical localization of
insulin in the chick embryo during development. Gen Comp Endocrinol, 73, 293-298, 1989.
24. Tomita T, Doull V, Pollock HG, Kimmell JR: Regional distribution
of pancreatic-polypeptide and other hormones in chicken pancreas - reciprocal relationship between pancreatic-polypeptide and glucagon. Gen Comp Endocr, 58, 303-310, 1985.
25. Boonen K BG, D’ Hertog W, Husson SJ, Overbergh L, Mathieu C, Schoofs L: Neuropeptides of the islets of langerhans: A peptidomics
study. Gen Comp Endocrinol, 152, 231-241, 2007.
26. Simsek N, Karadeniz A, Özüdogru Z, Kara A, Can I: An
immuno-histochemical study on the gastrin-, somatostatin-and serotonin-releasing cells in the gastrointestinal system of adult quails. Atatürk Üniv Vet Bil Derg, 6, 183-193, 2011.
27. Rawdon BB, Andrew A: An immunocytochemical study of the
distribution of pancreatic endocrine cells in chicks, with special reference to the relationship between pancreatic polypeptide and somatostatin-immunoreactive cells. Histochemistry, 59, 189-197, 1979.
28. Dockray GJ Varro A, Dimaline R, Wang T: The gastrins: Their
production and biological activities. Annu Rev Physiol, 63, 119-139, 2001.
29. Verme TB, Hootman SR: Regulation of pancreatic duct epithelial
growth in vitro. Am J Physiol, 258, 833-840, 1990.
30. Brand SJ, Fuller PJ: Differential gastrin gene expression in rat
gastrointestinal tract and pancreas during neonatal development. J Biol Chem, 263, 5341-5347, 1988.
31. Ding W-G, Fujimura M, Tooyama I, Kimura H: Phylogenetic study
of serotonin-immunoreactive structures in the pancreas of various vertebrates. Cell Tissue Res, 263, 237-243, 1991.
32. Takayanagi M, Watanabe T, Yamada J, Nagatsu I:
Immuno-cytochemical colocalizations of serotonin, aromatic L-amino acid de-carboxylase and polypeptide hormones in A- and PP-cells of the chicken endocrine pancreas. Tissue & Cell, 27, 439-446, 1995.