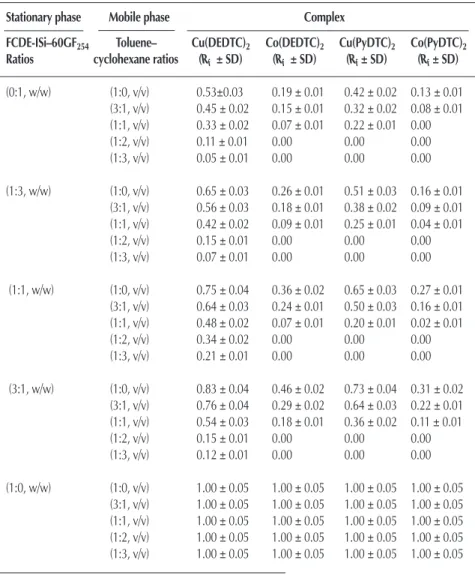
Acid modified diatomaceous earth - A sorbent material for thin layer chromatography
Tam metin
Şekil




Benzer Belgeler
Bu çalıĢma, derin öğrenme mimarisi olan evriĢimsel sinir ağı tabanlı yüz ayırt etme ve tanıma problemleri için iki yöntem önermektedir.. Uygulamamızda,
The first, second, and third parts are numerical fields, with the first part has the default value equal to the total number o f weekly hours, and the second and
We prove that the optimal solution to this problem corresponds to either (i) switching between at most two channels with deterministic signaling over each channel and employing
Before we proceed with our RHT0EA method, let us briefly describe two methods in literature for solving the higher level problem in (8); the forward decomposition (FWD) method, for
level with ( X :3,85) degree, “This game makes people addicted to play continuously.” item is on high “I agree” level with ( X :3,84) degree, “One’s enjoying a lot
Bilimselliğin ve liyakatın geleceğin adli bilimlerini inşa etmede tek gerçek olduğu bilinciyle dergimizin bilimsel niteliğini hep birlikte daha da yükseklere taşımak, Adli Tıp
We present a 29 weeks old 460 gr extremely low birth weight (ELBW) infant with congenital CMV infection mimicking total parenteral nutrition (TPN) associated cholestasis..
Çalışmamızda FMS hastalarda sesin aerodinamik parametrelerinden SPLmax da istatistiksel olarak anlamlı ancak SPLmin da istatistiksel olarak anlamlı olmayan bir azalma