Altitude training induced alterations in erythrocyte rheological properties: A controlled comparison study in rats
Tam metin
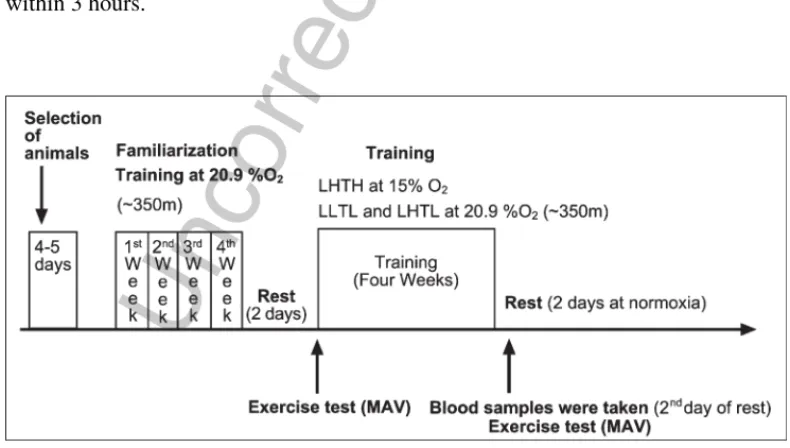
Şekil
Benzer Belgeler
Rekommendationerna kommer att speglas av genomförandet och resultaten från den nyligen avslutade så kallade OPEN-studien, där dagliga uppresningsövningar och dagligt
The anal fin of the male fish has evolved into a gonopodium, a stick-shaped organ used for reproduction. Platies are easy to keep and well suited to a
Live Earth İstanbul, ülkemizin bir seçim maratonunda olması, terör ve güvenlik nedenleriyle pek çok kişi ve kurumun maalesef önceliği olamadı" ifadelerine yer
Cultivation of microalgae in aquaculture are used directly for feeding mollusks, crustaceans and shelled larvae, but they are used as a first feed in indirect zooplankton feeding
This is a very optimistic result that can be a key for solving indoor blockage problem and allows either side of the connection to switch beams in the case of LOS blockage before a
The predictive modeling of the device requires a carefully parametrized the Ge and boron doping profiles. A thermodynamically stable box-like Ge profiles are included for simulation
Although inkjet bioprinting has been one of the most commonly used method 219 in printing living cells and biomaterials, cell aggregation, sedimentation and cell- 220
Artut, lisans derecesini Koç Üniversitesi’nde; yüksek lisansını Londra Middlesex Üniversitesi Ses Sanatları Bölümü’nde tamamladı ve doktorasını İsviçre’de