Başlık: Serum Cardiac Troponin-I in dogs with CPV-2 infection Yazar(lar):BASTAN, Idil; KURTDEDE, Arif; SEL, Tevhide; ÖZEN, Doğukan; YUMUŞAK, Nihat; TIMURKAN, M. Özkan; BAYDIN, Ahmet Cilt: 60 Sayı: 4 Sayfa: 251-255 DOI: 10.1501/Vetfak_0000002588 Yayın Tar
Tam metin
Şekil
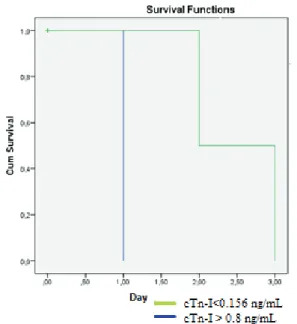
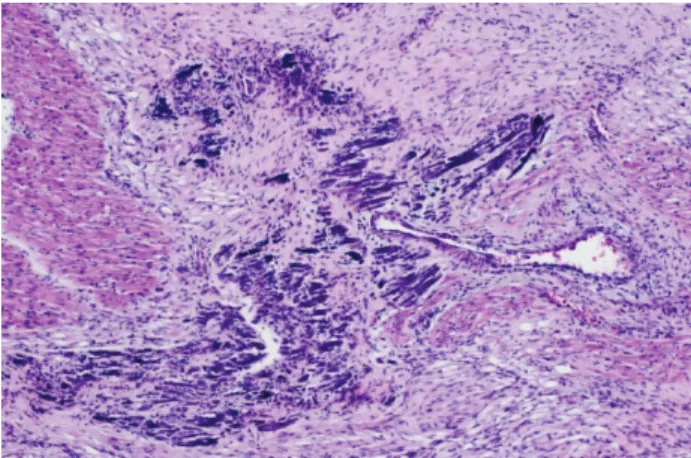
Benzer Belgeler
When, patients were subclassified according to extent of involvement of coronary artery territory as high (involvement of more than half of coronary tree) versus low stenosis
Objective: We investigated the association of serum asymmetric dimethylarginine (ADMA) with metabolic syndrome (MetS), type-2 diabetes and coronary heart disease (CHD) in the
應用校內研發能量,積極開創另頁的產學合作契機,落實產學互動,培育在校生的
The efficacy and safety of Q-switched potassium titanyl phosphate (KTP) and long-pulsed neodymium-doped yttrium aluminum garnet (lpNd:YAG) lasers were compared in the treatment
Similar findings were observed in a previous study investigating the link between periodontal and systemic diseases, revealing higher levels of salivary TREM-1 in chronic
Three transcription factors, Hnf4α, Foxa2 and Foxa3 has been transduced into MEF cells separately, and in combination of Hnf4α+Foxa2 and Hnf4α+Foxa3.[24] To best of our knowledge,
Denetleyici ve Düzenleyici Sistemler, vücudumuzda gerçekleşen olayların düzenli, birbiriyle uyumlu ve sorunsuz olması, hücrelerin bir araya gelerek
(Matthews, 1994). Aşağıdaki lemma ile arasındaki ilişkiyi vermekte olup sabit nokta çalışmaları için sonuç elde etmekte önemli bir rol oynar. kısmi metrik uzay olsun. ile