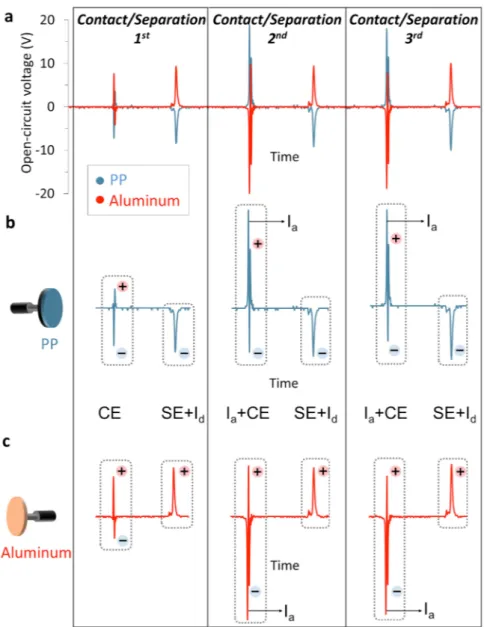
The charging events in contact-separation electrification
Tam metin
Şekil
Benzer Belgeler
The algorithm was implemented in the network simulator ns-2 so that a large number of experiments could be performed to assess its accuracy and effect on server (node which
Tenâsüh inancı İslâm filozofları nezdinde de kabul görmemiştir. Ancak filo- zoflar tenâsühü red konusunda âhiret inancından daha ziyâde, rûhun ferdîliğini esas alan
Ebeveynlerin Çocukluk Aşılarına Yönelik Tutumları Ölçeğinin (PACV) orijinal sürümünün üç boyutlu yapısının Türkçe sürüm ile uyumunu gösteren
Öğretmen Motivasyonunu, Boyutların Toplam Puanları Üzerinden, Yordamasına İlişkin Yol Analizi Sonuçları.. Bunun yanında CFI, NFI, IFI, SRMR ve RMSEA kabul
Since optical systems consisting of arbitrary concatena- tions of any number of sections of free space and lenses can be mathematically expressed as linear canonical transforms,
KC hemanjiomları potansiyel olarak ani ölümlere sebep olabilen spontan rüptür ve kanama riskine sahiptirler, hemanjiomların rüptür ihtimali %1-4 gibi düşük olmasına
Separations can be achieved by differences in physical properties, such as differences in boiling point, or by chemical means, wherein differences in physical properties are
The single-shell cell models are widely employed mathematical models for dielectrophoresis owning to their simplicity and fast computation costs, Fig. Dielectric