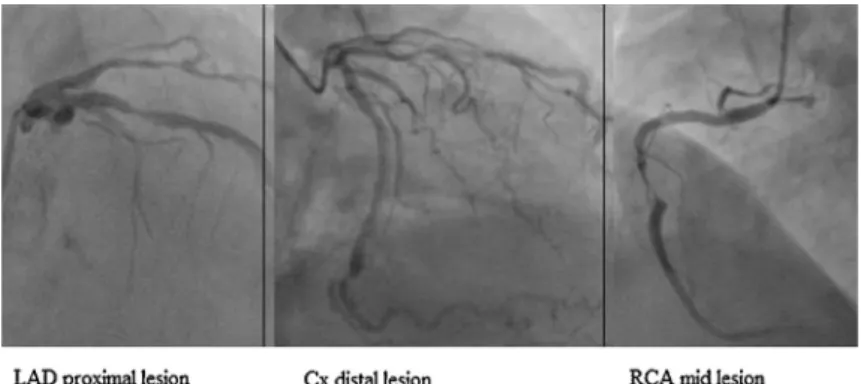
Evans syndrome with non-ST segment elevation myocardial infarction complicated by hemopericardium
Tam metin
Şekil

Benzer Belgeler
The present study, which is the first epidemiologic study investigating risk factors for STEMI in Turkish population, showed that the potentially modifiable risk factors,
Although the conduction abnormalities of the left anterior and posterior fascicles, left anterior fascicular block (LAFB), and left posterior fascicular block (LPFB) are
Objective: This study aims to investigate the association of circulating miR-660-5p with no-reflow phenomenon (NRP) in patients with ST segment elevation myocardial infarction
The objective of the present study is to figure out the relation between admission sK level and in-hospi- tal mortality, ventricular arrhythmias, long-term (six months) mortality,
The investigators reported that admission serum potassium (sK) level of >4.5 mmol/L was associated with increased long-term mortality, and significant relation was de-
The investigators reported that admission serum potassium (sK) level of >4.5 mmol/L was associated with increased long-term mortality, and significant relation was de-
In this study, we examined the expression levels of miRNA related to cardiac diseases in circulating blood among STEMI patients versus a control group to identify miRNA
Considering these results, we suggest that hepcidin could not be use as a marker of myocardial necrosis in acute phase such as troponin in patients with NSTEMI, since increasing