Myrtus communis L.: Characterisation of Essential Oil of Leaves and Fatty Acids of Seeds Using Gas Chromatography-Mass Spectrometry (GC/MSD)
Tam metin
Şekil
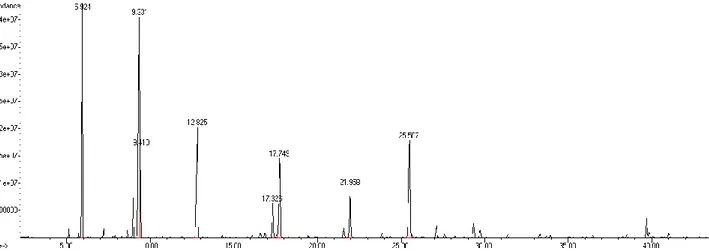
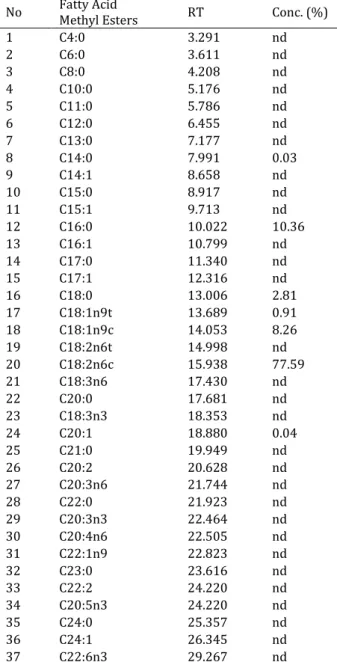
Benzer Belgeler
Bu çalışmanın bulguları da, Çim Biçme adlı modelleme probleminde Grup1 ve Grup 2’deki öğrencilerin kendi matematiksel fikirlerini puanlama olarak geliştirdiklerini ve
Şöyle ki, ondokuzuncu yüzyılın ikinci yarısında, kolonyalizm=emperyalizm ça- ğında Osmanlı siyasi-idari literatürüne “adem-i merkeziyet” kavramın girmesi ve
That is, the adversary with the knowledge that the target dataset has dependent tuples can infer more SNPs as the number of family members included in the query results increases from
One of the more colourful episodes in the relations of tenth-century English kings with their Celtic neighbours is the submission of various Scottish, Welsh and Scandinavian rulers
We believe that the evidence in the literature—which links the strength of social ties, the level of disagreement, the social context of network ties, and the political sophisti-
In order to do this we followed the paradigm of [34]. In their study, they ad- ministered an animal detection task and concluded that shape and texture are the most effective cues
İslam düşüncesinde, pek çok eserde bahsi geçen farklı bilgilerden Hermes’in tanındığı anlaşılmaktadır. Biz bu yüksek lisans tezinde Hermes’in İslam
Son yıllarda, Türk Milli Eğitim Sisteminde okul öncesi eği tim, altı yaş grubu çocukların eğitimi, özel eğitim, yaygın eği tim gibi olguların ele