Injury, Int. J. Care Injured 50 (2019) 1915–1920
Effect
of
curcumin
on
bone
healing:
An
experimental
study
in
a
rat
model
of
femur
fracture
Selim
Safali
a,
Bahattin
Kerem
Aydin
a,*
,
Alaaddin
Nayman
b,
Ceyhan
Ugurluoglu
c aSelcukUniversity,DepartmentofOrthopaedicsandTraumatology,TurkeybSelcukUniversity,DepartmentofRadiology,Turkey cSelcukUniversity,DepartmentofPathology,Turkey
ARTICLE INFO Articlehistory: Accepted1September2019 Keywords: Rat Femur Fracturehealing Curcumin ABSTRACT
Objective:Todeterminetheradiologic,histologicandbiomechanicaleffectsofcurcuminonbonehealing
usingatotalratfemurfractureinjurymodel.
Materialsandmethods:SixtyfourmaleWistar–Albinoratsweighing170–210gwereusedinthisstudy.
Theanimalswererandomlydividedintoeightgroupsand5or6animalswereplacedineachcage.A
transversefemurshaftfracturemodelused.Theanimalsinstudygroupsreceivedoralcurcuminatadose
of200mg/kgfor14daysor28days.Remaininganimalsreceivedonlysalinesolutionbyoralgavagefora
periodof14daysand28daysascontrolgroups.Aftersacrificationtheleftfemursusedforradiological,
histologicalandbiomechanicalevaluation.
Results: The groupstreated withcurcuminshowed nosignificant differencein termsof radiological,
histologicalandbiomechanicalevaluationsin14daysgroups.Alsotherewasnosignificantdifferencebetween
curcuminandcontrolgroupsfor28daysaccordingtoradiological,histologicalandbiomechanicaltests.
Conclusions:Accordingtoourresults,curcuminhasnopositiveeffectonfracturehealingnotonly
histologicallybutalsoradiologicallyandbiomechanically.Curcumin’santioxidanteffectmaybemore
noticeablewithlongtermfollowupinvestigationasitmayhaveapositiveeffectinremodellingphase.
Longtermfollowupdesignedstudiesmaybeplannedtoinvestigateitseffectonremodellingphaseof
fracturehealing.
©2019ElsevierLtd.Allrightsreserved.
Introduction
Fracture healing continues to be a significant challenge for orthopaedic surgeons. It leads to significant health care costs which resultin restrictedactivityand individualmorbidity [1]. Boneisauniquetissuewithastronghealingcapabilityintermsof restoringoriginaltissuebutnotintermsoffibrosisrepair.Healing isacomplexandorderlyregulatedprocessthatcanbeaffectedby multiplefactors.However,previouspublicationshaveshownthat in5% 10%ofbonefractures,healingcannotbeachievedleadingto impairedqualityoflife[1,2].
Curcumin, a natural compound extracted from turmeric (Curcuma longa), has strongantioxidant and anti-inflammatory activities.Morethan10,000articleshavediscussedthemolecular
basisofcurcumin’spotentialantioxidant,anti-inflammatory,and anti-infectious activities during the last decade [3–7]. Many studieshaveshowncurcumin’spositiveeffectsontissueinjuries ofthedermis,nervefibres,andburnsaremediatedviaactivationof cellmigrationandwoundhealing[8–10].Ithasbeenreportedthat antioxidanttherapyandinhibitinginflammatoryprocessescould havepositiveeffectsonthehealingofsofttissueinjuries[1,11–13]. Manyfactorshavebeenstudiedfortheirroleinimprovingbone healing processes such as stem cells, growth factors, and haemostaticagents[14–17]. However,untilnow,therehasbeen only one reported study in the English literature assessing curcumin’seffectonbonehealingviahistologicandradiological evaluationbutnotbiomechanicalevaluation[18].
In thepresentstudy,weaimedtoinvestigatetheradiologic, histologic,andbiomechanicaleffectsofcurcuminonbonehealing usingaratfemurtotalfractureinjurymodel.
Materialsandmethods
Sixtyfour maleWistar albinoratsweighing 170–210gwere used.Theexperimentaldesignandallprocedureswereapproved
* Correspondingauthorat:SelcukUniversity,FacultyofMedicine,Departmentof Orthopaedicsand Traumatology, AlaeddinKeykubat Campus,42100, Selcuklu, Konya,Turkey.
E-mailaddresses:[email protected](S.Safali),[email protected]
(B.K.Aydin),[email protected](A.Nayman),[email protected]
(C.Ugurluoglu).
https://doi.org/10.1016/j.injury.2019.09.002
0020-1383/©2019ElsevierLtd.Allrightsreserved.
Injury,Int.J.CareInjuredxxx(2019)xxx–xxx
GModel
JINJ8304No.ofPages6
ContentslistsavailableatScienceDirect
Injury
1916 S. Safali et al. / Injury, Int. J. Care Injured 50 (2019) 1915–1920
byXXXAnimalResearchandEthicsCommittee(protocolnumber 2015/38).Alltheanimalsforthisstudywerepurchasedfromthe LaboratoryofExperimentalAnimals,XXXUniversity,BBcity,CCC Country.
Theanimalswererandomlydividedintoeightgroupsandfive orsixanimalswereplacedineachcage. Theeightgroupswere designatedas A,B, C, D,E,F,G,and H.Theproperties andthe numbersoftheanimalsofeachgroupareshowninTable1.
A well-known rat fracture model was used [18]. Surgical procedures were conducted under general anaesthesia with intraperitoneal injection of 50mg/kg ketamine (Ketalar; Eczacıbaşı, _Istanbul,Turkey)and10mg/kgxylazinehydrochloride (Rompun;Bayer,Leverkusen,Germany).Theleftlowerlimbsinthe rats were shaved and disinfected using povidone-iodine, and surgerywas performedundersterile conditions.A 3-cmlateral longitidinal incisions were made to expose the femurs. A transverse femur shaft fracture was made using an ossilating microsaw.A1-mmKirschnerwire(Hipokrat, _Izmir,Turkey)was usedforintramedullaryfixation.Thesubcutaneoustissueandthe skinwereclosedfollowingstandardprocedures.
Nobandageorcastingwasplacedafterthesurgeries.Allrats wereallowedtomovefreelyandwerefedwithstandardlaboratory feedandtapwater.
TheanimalsingroupsAandCreceivedcurcumin(curcumin, Sigma-Aldrich,Germanysuspendedinsalineatadoseof200mg/ kgorally)viaoralgavagefor14daysandtheanimalsinGroupE andFreceivedcurcuminforfourweeksstartingfromthedayof surgery.Theremaininganimalsreceivedonlysalinesolutionvia oralgavageforaperiodof14days(ingroupsBandD)or28days(in groupsEandF).Thedosageofcurcuminwasbasedonprevious similarstudies[13,19].
High-doseethervapourwasusedforeuthanasia.Aftertherats werekilled,theirleftfemursweredisarticulatedfromtheirhipand kneejoints.Softtissuesonthefemoralbonewerepeeledoffgently fromthebonewithoutanyharmtothecallustissue.Allofthe left femurs were studied addressing clinical, radiological, and histologicalaspects.
Alltheleftfemurswereremovedandtakenimmediatelyfor radiologicanalysis.ThefemursobtainedfromgroupsA,B,E,andF
were used for histological analysis. The femurs obtained from groupsC,D,G,andHwereusedforbiomechanicalanalysis. Radiologicanalysis
Sixtyfourfemurswereusedforradiologicalanalysis.Computed tomography (CT) imaging was performed with a 128 2-slice dual-sourceCT (Somatom Definition Flash, Siemens, Germany). ThesampleswereplacedintheCTforobtainingmicro–CTimages byscanningalong thelongaxisof thefemoralsamplesandthe regionofinterest,10mmaroundthefracturesite,witha0.6mm collimation.TheKwireswerenotremovedbecausetheydidnot causeanyartefactsinradiologicalanalysis.
The software Radiant Dicom Wiever 4.6.9 was used for radiologicalmeasurements.Totalcallusdiameter,low-radiodensity bonemeasurements,highdensitybonemeasurements,lengthof callustissue,andfemoraldiameterparametersweremeasured.For measuringcallusdiameter,low-radiodensitybonemeasurements, high density bone measurements, and femoral diameter, axial sectionswereused.Analysisaftertwoweekswasdoneusingthe ratiooftotalcallusdiameter/femoraldiameter,andanalysisafter fourweekswas doneusingtheratio oflowdensitybone/highdensity bonemeasurements[Fig.1].
Histologicalanalysis
Atotal 32 femursfromgroups A,B, D,and Ewereusedfor histologicalanalysis. A bufferedformalin solutionwas usedfor fixationovertwodays.Next,10%aceticacidsolutionwasusedfor decalcificationoverfourdays.Next,thespecimenswere embed-dedinparaffinand3mm-thickserialsectionsweretakenfromthe callus site of each femur. Hematoxylin and eosin staining was performed.Histologicalclassificationofthehealingwasdoneper thehistologicalhealingscalepublishedbyHuoetal.[20]. Biomechanicalanalysis
Atotal32leftfemursoftheratsfromgroupsC,D,G,andHwere used for biomechanical analysis. Biomechanical testing were
Table1
Thedistrubutionofgroups,contentsandsacrificationtimes.
GroupName GroupFeature NumberofRats SacrificationTime
A 14daysHistologicalAssesmentCurcuminGroup 10 14days
B 14daysHistologicalAssesmentControlGroup 6 14dayd
C 14daysBiomechanicalAssesmentCurcuminGroup 10 14days
D 14daysBiomechanicalAssesmentControlGroup 6 14days
E 28daysHistologicalAssesmentCurcuminGroup 10 28days
F 28daysHistologicalAssesmentControlGroup 6 28days
G 28daysBiomechanicalAssesmentCurcuminGroup 10 28days
H 28daysBiomechanicalAssesmentControlGroup 6 28days
Fig.1.Schematicdrawingsofmeasurementsusedforradiologicalevaluation.A,longitudinalsection.B,Transversesection.
2 S.Safalietal./Injury,Int.J.CareInjuredxxx(2019)xxx–xxx
S. Safali et al. / Injury, Int. J. Care Injured 50 (2019) 1915–1920 1917
byXXXAnimalResearchandEthicsCommittee(protocolnumber 2015/38).Alltheanimalsforthisstudywerepurchasedfromthe LaboratoryofExperimentalAnimals,XXXUniversity,BBcity,CCC Country.
Theanimalswererandomlydividedintoeightgroupsandfive orsixanimalswereplacedineachcage. Theeightgroupswere designatedas A,B, C, D,E,F,G,and H.The propertiesand the numbersoftheanimalsofeachgroupareshowninTable1.
A well-known rat fracture model was used [18]. Surgical procedures were conducted under general anaesthesia with intraperitoneal injection of 50mg/kg ketamine (Ketalar; Eczacıbaşı, _Istanbul,Turkey)and10mg/kgxylazinehydrochloride (Rompun;Bayer,Leverkusen,Germany).Theleftlowerlimbsinthe rats were shaved and disinfected using povidone-iodine, and surgerywas performedunder sterileconditions.A 3-cmlateral longitidinal incisions were made to expose the femurs. A transverse femur shaft fracture was made using an ossilating microsaw.A1-mmKirschnerwire(Hipokrat, _Izmir,Turkey)was usedforintramedullaryfixation.Thesubcutaneoustissueandthe skinwereclosedfollowingstandardprocedures.
Nobandageorcastingwasplacedafterthesurgeries.Allrats wereallowedtomovefreelyandwerefedwithstandardlaboratory feedandtapwater.
TheanimalsingroupsAandCreceivedcurcumin(curcumin, Sigma-Aldrich,Germanysuspendedinsalineatadoseof200mg/ kgorally)viaoralgavagefor14daysandtheanimalsinGroupE andFreceivedcurcuminforfourweeksstartingfromthedayof surgery.Theremaininganimalsreceivedonlysalinesolutionvia oralgavageforaperiodof14days(ingroupsBandD)or28days(in groupsEandF).Thedosageofcurcuminwasbasedonprevious similarstudies[13,19].
High-doseethervapourwasusedforeuthanasia.Aftertherats werekilled,theirleftfemursweredisarticulatedfromtheirhipand kneejoints.Softtissuesonthefemoralbonewerepeeledoffgently fromthebonewithoutanyharmtothecallustissue. Allof the left femurs were studied addressing clinical, radiological, and histologicalaspects.
Alltheleftfemurswereremovedandtakenimmediatelyfor radiologicanalysis.ThefemursobtainedfromgroupsA,B,E,andF
were used for histological analysis. The femurs obtained from groupsC,D,G,andHwereusedforbiomechanicalanalysis. Radiologicanalysis
Sixtyfourfemurswereusedforradiologicalanalysis.Computed tomography (CT) imaging was performed with a 128 2-slice dual-source CT (Somatom Definition Flash, Siemens, Germany). ThesampleswereplacedintheCTforobtainingmicro–CTimages byscanningalongthelongaxisofthefemoralsamplesandthe regionofinterest,10mmaroundthefracturesite,witha0.6mm collimation.TheKwireswerenotremovedbecausetheydidnot causeanyartefactsinradiologicalanalysis.
The software Radiant Dicom Wiever 4.6.9 was used for radiologicalmeasurements.Totalcallusdiameter,low-radiodensity bonemeasurements,highdensitybonemeasurements,lengthof callustissue,andfemoraldiameterparametersweremeasured.For measuringcallusdiameter,low-radiodensitybonemeasurements, high density bone measurements, and femoral diameter, axial sectionswereused.Analysisaftertwoweekswasdoneusingthe ratiooftotalcallusdiameter/femoraldiameter,andanalysisafter fourweekswas doneusingtheratio oflowdensitybone/highdensity bonemeasurements[Fig.1].
Histologicalanalysis
Atotal 32 femursfromgroups A,B, D,and Ewereusedfor histologicalanalysis. A bufferedformalin solutionwas usedfor fixationovertwodays.Next,10%aceticacidsolutionwasusedfor decalcificationoverfourdays.Next,thespecimenswere embed-dedinparaffinand3mm-thickserialsectionsweretakenfromthe callus site of each femur. Hematoxylinand eosin staining was performed.Histologicalclassificationofthehealingwasdoneper thehistologicalhealingscalepublishedbyHuoetal.[20]. Biomechanicalanalysis
Atotal32leftfemursoftheratsfromgroupsC,D,G,andHwere used for biomechanical analysis. Biomechanical testing were
Table1
Thedistrubutionofgroups,contentsandsacrificationtimes.
GroupName GroupFeature NumberofRats SacrificationTime
A 14daysHistologicalAssesmentCurcuminGroup 10 14days
B 14daysHistologicalAssesmentControlGroup 6 14dayd
C 14daysBiomechanicalAssesmentCurcuminGroup 10 14days
D 14daysBiomechanicalAssesmentControlGroup 6 14days
E 28daysHistologicalAssesmentCurcuminGroup 10 28days
F 28daysHistologicalAssesmentControlGroup 6 28days
G 28daysBiomechanicalAssesmentCurcuminGroup 10 28days
H 28daysBiomechanicalAssesmentControlGroup 6 28days
Fig.1.Schematicdrawingsofmeasurementsusedforradiologicalevaluation.A,longitudinalsection.B,Transversesection.
2 S.Safalietal./Injury,Int.J.CareInjuredxxx(2019)xxx–xxx
GModel
JINJ8304No.ofPages6
performedusingaElistaTST2500materialtestingmachine(Elista, Istanbul,Turkey).Thefemursweremountedwithminiclampson thetestingdevice.Theclampsgrippedeachspecimenatthedistal and proximal metaphyseal parts of the femurs for measuring resistance to longitudinal distraction forces (in Newtons). A distractionforcewasappliedat3mm/min.Thedistractionforced appliedwasincreaseduntilfailureofthehealingregion.Loadto failureparameterswererecordedforeachspecimen.Athree-point bendingtestwasplannedbutcouldnotbeusedbecausetwo-week specimenswerenotusableduetoonlysoftcallusformation.The recordedmeasurements(inNewtons)wereusedtocomparethe resultsofbiomechanicalevaluation.
Statisticalanalysis
Mann–Whitney U test was used for analysing radiological, histological, and biomechanical results to evaluate differences betweenthetwogroups. Alltheanalyseswereperformedwith IBMSPSSversion 20.0(IBMCorp., Armonk,NY,USA.A p value <0.05wasconsideredtobestatisticallysignificant).
Results
RadiologicalresultsbasedonCTimagesareshowninTable2. There was no significant difference in radiological evaluation resultsbetweenthecurcumin2nd weekgroupandthecontrol group (Mann–Whitney test; U=117.00; p>0.05) [Fig. 2]. For radiologicalevaluationaftertwoweeks,onlytotalcallusdiameter/ femoraldiameter measurement wereused becausehard callus formationwasinsufficient.Comparisonofradiologicalevaluation resultsfromtheratskilledinthesecondweekshowedthatthe
diameterofthecallusinthecurcumingroupwassmallerthanthat inthecontrolgroup.Atfourweeks,theratiooflowdensitybone/ highdensitybonewasusedtorepresenthealing.Lowdensitybone is a predictor of new bone formation and high density bone indicates the cortex of the intact (before fracture) bone. Radiological evaluation results of the rats killed in the fourth weekshowed thatthelow-radiodensity bone/highradiodensity bone ratiowas higherinthecurcumingroup[Fig.3].However, statisticalcomparisonofradiologicalevaluationresultsattwoor fourweeksdidnotshowanystatisticalsignificance.
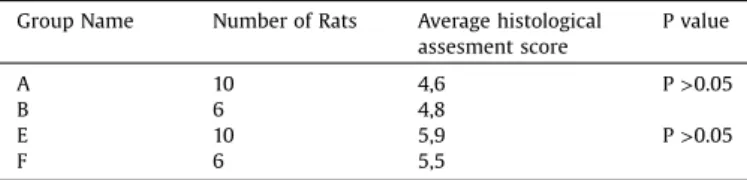
Histological scores per the Huo classification are shown in
Table 3. There was no significant difference in histological examinationresultsbetweenthe2ndweekcurcumingroupand the 2ndweekcontrol group(Mann–WhitneyU test; U=30.00; p>0.05)[Fig.4].Therewasnosignificantdifferenceinhistological examinationresultsbetweenthe4thweek curcumingroupand 4th week control group (Mann–Whitney U test; U=21.00; p>0.05) [Fig. 5]. The mean scores of histological examination were4.8inthecontrolgroupand4.2inthecurcumingroupattwo weeks.Themeanscoreswere5.5inthecontrolgroupand5.9inthe curcumingroupatfourweeks.Thescoresattwoweekswerebetter inthecontrolgroup,andthoseatfourweekswerebetterinthe curcumingroup;however,thesedifferenceswerenotstatistically significant.
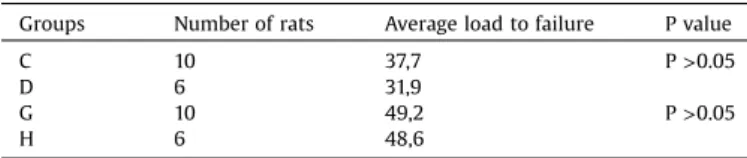
The results of biomechanical analysisare shown inTable 4. Therewasnosignificantdifferenceinbiomechanicalexamination resultsbetweenthe2ndweekcurcumingroupandthe2ndweek controlgroup(Mann–WhitneyUtest;U=29.00;p>0.05).There was nosignificantdifference inthebiomechanical examination resultsbetweenthe4thweekcurcumingroupandthe4thweek control group (Mann–Whitney U test; U=27.50; p>0.05). The meanloadtofailurewas8.3Ninthecontrolgroupand8.6Ninthe curcumingroupaftertwoweeks;itwas8.9inthecontrolgroup and8.2inthecurcumingroupafterfourweeks.
Discussion
There are limited studies on curcumin’s effect on fracture healing.Curcumin’spositiveeffectonfracturehealinghasbeen reported in a recent study, which found that curcumin has potentialinpromotingbonehealingbyactivatingautophagyina
Table2
Radiologicalresultsofcurcuminandcontrolgroups. Groups Numberofrats Averageradiological
assesmentscore Pvalue A+C 20 2,514 P>0.05 B+D 12 2,509 E+G 20 1,314 P>0.05 F+H 12 1,281
Fig.2.A,Sampleoftransverseradiologicalimaging(micro-CT)afterscarificationat14daysfromCurcumingroup.B,Sampleoftransverseradiologicalimagingafter scarificationat14daysfromcontrolgroup.
S.Safalietal./Injury,Int.J.CareInjuredxxx(2019)xxx–xxx 3
GModel
1918 S. Safali et al. / Injury, Int. J. Care Injured 50 (2019) 1915–1920
ratmodel [18]. Histologicaland radiologicalevaluation showed positiveeffectsof curcuminonbonehealingbut biomechanical tests were not used in the above study. To the best of our knowledge, the present study is the first animal model study investigatingcurcumin’seffectonfracturehealingusing radiolog-ical,histological,andbiomechanicaltests.
Inthepresentstudy,wecreatedafemurfractureinjurymodel andevaluatedtheeffectsoforalcurcumin.Thehistologicalresults werebetterin thecurcumingroupattwo andfourweeks, but the differences were not statistically significant. This may be secondarytocurcumin’santi-inflammatoryeffect.Thefirstphase
of bone healing is theinflammation phase and curcumin may inhibit this phase. A similarsituation mayexistat fourweeks, representingthehealingorcallusphaseinfracturehealing.Thisis quitedifferentfrompreviouslyreportedfindings[18].Theirstudy designisquitedifferentfromourstudy.Firstly,theyusedalongK wirefixationtechniquepenetratingthekneejointandendsoutof theskin.Thistypeoffixationcanblockthemotionofthekneejoint andloadoftheextremitywhichmaybepositivefactorforfracture healing.InourstudyweusedshortKwireswhichwerecompletely buriedintramedullaryfornotlimitingtheearlyloadandrangeof motionofthejoints.Secondlythetimeperiodisforsacrifyingthe animalsisdifferentastheyusedtwoandsixweeksresults.Thirdly, Lietal.designedtheirstudyonblockingtheautophagyoftherats byusing3-methyladenineandcurcuminbuttheydidnotstudythe only effect of 3-methyladenine on fracture healing. Also the histologicalevaluationsysteminourstudywasdifferentfromthat usedintheabovestudy.Inourstudy,weevaluatedhealingusing Huo’s classification, which has 10parameters, using only light microscopywithhaematoxylinandeosinstaining.Theabovestudy usedosteoblastcellcounting,immunohistochemicaltissue stain-ing,andwesternblotting.However,thereisnoreportedobjective
Fig.3.A,Sampleoflongitudinalradiologicalimaging(micro-CT)afterscarificationat28daysfromcurcumingroup.B,Sampleoflongitudinalradiologicalimagingafter scarificationat28daysfromcontrolgroup.
Table3
Histologicalexaminationresultsofcontrolgroupandcurcumingroup. GroupName NumberofRats Averagehistological
assesmentscore Pvalue A 10 4,6 P>0.05 B 6 4,8 E 10 5,9 P>0.05 F 6 5,5
Fig.4.A,Histologicalimagesofhealingaftertwoweeks(H&EstainingX100).A,Curcumingroupshowingmainlychondroidtissueandsomewovenbone.B,Controlgroup showingequalamountofchondroidtissueandwovenbone.
S. Safali et al. / Injury, Int. J. Care Injured 50 (2019) 1915–1920 1919
ratmodel [18]. Histologicaland radiologicalevaluation showed positiveeffectsof curcuminonbonehealingbut biomechanical tests were not used in the above study. To the best of our knowledge, the present study is the first animal model study investigatingcurcumin’seffectonfracturehealingusing radiolog-ical,histological,andbiomechanicaltests.
Inthepresentstudy,wecreatedafemurfractureinjurymodel andevaluatedtheeffectsoforalcurcumin.Thehistologicalresults werebetterin thecurcumingroupattwo andfourweeks, but the differences were not statistically significant. This may be secondarytocurcumin’santi-inflammatoryeffect.Thefirstphase
of bone healing is the inflammationphase and curcumin may inhibit this phase. A similarsituation mayexistat four weeks, representingthehealingorcallusphaseinfracturehealing.Thisis quitedifferentfrompreviouslyreportedfindings[18].Theirstudy designisquitedifferentfromourstudy.Firstly,theyusedalongK wirefixationtechniquepenetratingthekneejointandendsoutof theskin.Thistypeoffixationcanblockthemotionofthekneejoint andloadoftheextremitywhichmaybepositivefactorforfracture healing.InourstudyweusedshortKwireswhichwerecompletely buriedintramedullaryfornotlimitingtheearlyloadandrangeof motionofthejoints.Secondlythetimeperiodisforsacrifyingthe animalsisdifferentastheyusedtwoandsixweeksresults.Thirdly, Lietal.designedtheirstudyonblockingtheautophagyoftherats byusing3-methyladenineandcurcuminbuttheydidnotstudythe only effect of 3-methyladenine on fracture healing. Also the histologicalevaluationsysteminourstudywasdifferentfromthat usedintheabovestudy.Inourstudy,weevaluatedhealingusing Huo’s classification, which has 10parameters, using only light microscopywithhaematoxylinandeosinstaining.Theabovestudy usedosteoblastcellcounting,immunohistochemicaltissue stain-ing,andwesternblotting.However,thereisnoreportedobjective
Fig.3.A,Sampleoflongitudinalradiologicalimaging(micro-CT)afterscarificationat28daysfromcurcumingroup.B,Sampleoflongitudinalradiologicalimagingafter scarificationat28daysfromcontrolgroup.
Table3
Histologicalexaminationresultsofcontrolgroupandcurcumingroup. GroupName NumberofRats Averagehistological
assesmentscore Pvalue A 10 4,6 P>0.05 B 6 4,8 E 10 5,9 P>0.05 F 6 5,5
Fig.4.A,Histologicalimagesofhealingaftertwoweeks(H&EstainingX100).A,Curcumingroupshowingmainlychondroidtissueandsomewovenbone.B,Controlgroup showingequalamountofchondroidtissueandwovenbone.
4 S.Safalietal./Injury,Int.J.CareInjuredxxx(2019)xxx–xxx
GModel
JINJ8304No.ofPages6
classificationsystemforthishistologicexamination.Indeed,the number ofosteoblasts and otherimmunohistochemical param-etersareimportantinfracturehealing,butwecouldnotfindany reportscomparingHuo’shistologicclassificationmethodandthe osteoblastcountingmethod.Itisknowninsomecases,although thenumberofblasticcellsishigh,itmaynotbefunctional(suchas hypertrophicpseudoarthrosis).Therefore,thedifferencein histo-logicalresultsbetweenthesesimilarstudiesmaybesecondaryto theusageofdifferenthistologicalevaluationmethods.
Theradiologicalevaluationresultswerebetterinthecurcumin group,whichalsodidnotshowstatisticallysignificantdifferences. CTevaluationwas used for investigatingbony union in therat femurs.Thediameterofcallusformationanditsratiowithintact bonediameterwasusedforradiologicexaminations.Radiological evaluationresultsfrom similarreports haveshown statistically betterresultswithcurcuminuseinX-rayvisualisation;onlythe callusdiameterwasusedasaparameterintheirCTevaluation.In ourstudy,weusedcallusdiameterpluscallusdiametertointact bone ratio. Thedifference in ourCT evaluation results may be secondarytothissituation.
Wealsoperformedanalysisofbiomechanical evaluation;the maximum load to failure parameter was measured in all rats. Three-pointbendingtestscouldnotbeappliedbecausethecallus tissuesweretoosoft.Allthefailureswereatthefracturehealing site;thisissimilartothatseeninhumanre-fractures.The two-weekresultswerebetterinthecurcumingroupandthefour-week results were better in the control group. However, when we comparedtheresults,therewasnostatisticallydifferencebetween thecontrolandcurcumingroups.Therehasbeenonlyonereport onthebiomechanicaleffectsofcurcuminonbonetissue[21];in theirstudy,theauthorsevaluatedtheeffectofcurcuminonintact bonetissuebyusinganovariectomisedratmodel.Theyconcluded thatcurcuminhadnostatisticallypositiveeffectonbonetissuevia evaluationofbiomechanicaltests.Ourresultsaresimilartothatof theabovestudy,althoughweusedafemurfracturemodel.
Therearealsosomelimitationsinourstudy.First,wecouldnot applythethree-pointbendingtestforbiomechanicalevaluation.
Althoughfracture-healedtissueswerenotapplicableforthistest, longer follow-up analysis can be performed to address this. Secondly,thenumberof ratsusedwould needtobehigher for moreaccurateresults.
Curcuminhasbeenused,especiallyinAsiancountries,totreat differentkindsofdiseasesduetoitsantioxidant, anti-inflamma-tory,immunomodulatory,andantimicrobialeffectsformanyyears [3–10]. In ourstudy, we found that curcumindidnot improve fracture healing based on biomechanical, radiological, and histologicalevaluation.Weinferthatcurcumin’s anti-inflamma-torypropertymayberesponsibleforitsineffectivenessinfracture healing.
Conclusion
Our resultsshowed thatcurcumin hasnopositive effecton fracturehealing,notonlyhistologicallybutalsoradiologicallyand biomechanically. Curcumin’s effect may be more noticeable in long-term follow-up investigations because of its potential positive effects in the remodelling phase. Long termfollow up designed studies may be planned to investigate its effect on remodellingphaseoffracturehealing.
Statementofwelfareofanimals
Allapplicableinternational,national andinstitutional guide-linesforthecareanduseofanimalswerefollowed.Allprocedures performed were in accordance with ethical standards of the institution (Selcuk University Experimental Research Centre, Konya,Turkey)wherethestudywasconducted.
Acknowledgement
ThisstudywasfundedbySelcukUniversityScientificResearch Office.
References
[1]BuzaJR,EinhornT. Bonehealingin2016. ClinCases MinerBoneMetab 2016;13:101–5.
[2]SongY,HanGX,ChenL,ZhaiYZ,DongJ,ChenW,etal.Theroleofthe hippocampusandthefunctionofcalcitoningene-relatedpeptideinthe mechanismoftraumaticbraininjuryacceleratingfracture-healing.EurRev MedPharmacolSci2017;21:1522–31.
[3]ZhangW,LiX,ComesFranchiniM,XuK,LocatelliE,MartinRC,etal.Controlled releaseofcurcuminfromcurcumin-loadednanomicellestoprevent peritendinousadhesionduringAchillestendonhealinginrats.IntJNanomater 2016;11:2873–81.
Fig.5.A,Histologicalimagesofhealingafterfourweeks(H&EstainingX100).A,Curcumingroupshowingmainlywovenboneandsomechondroidtissue.B,Controlgroup showingmainlychondroidtissueandsomewovenbone.
Table4
Biomechanicalexaminationresultsofcurcuminandcontrolgroups.
Groups Numberofrats Averageloadtofailure Pvalue
C 10 37,7 P>0.05
D 6 31,9
G 10 49,2 P>0.05
H 6 48,6
S.Safalietal./Injury,Int.J.CareInjuredxxx(2019)xxx–xxx 5
GModel
1920 S. Safali et al. / Injury, Int. J. Care Injured 50 (2019) 1915–1920
[4]HeY,YueY,ZhengX,ZhangK,ChenS,DuZ.Curcumin,inflammation,and chronicdiseases:howaretheylinked?Molecules2015;20:9183–213. [5]StrimpakosAS,SharmaRA.Curcumin:preventiveandtherapeuticproperties
inlaboratorystudiesandclinicaltrials.AntioxidRedoxSignal2008;10:511–46. [6]LiX,ChenS,ZhangB,LiM,DiaoK,ZhangZ,etal.Insituinjectable nano-compositehydrogelcomposedofcurcumin,N,O-carboxymethylchitosanand oxidizedalginateforwoundhealingapplication.IntJPharm2012;437:110–9. [7]YuL,YiJ,YeG,ZhengY,SongZ,YangY,etal.Effectsofcurcuminonlevelsof nitricoxidesynthaseandAQP-4inaratmodelofhypoxia-ischemicbrain damage.BrainRes2012;1475:88–95.
[8]KulacM,AktasC,TulubasF,UygurR,KanterM,ErbogaM,etal.Theeffectsof topicaltreatmentwithcurcuminonburnwoundhealinginrats.JMolHistol 2013;44:83–90.
[9]Ma J, Liu J, YuH,WangQ, Chen Y,Xiang L. Curcuminpromotesnerve regenerationandfunctionalrecoveryinratmodelofnervecrushinjury. NeurosciLett2013;547:26–31.
[10]CastangiaI,NácherA,CaddeoC,ValentiD,FaddaAM,Díez-SalesO,etal. Fabricationofquercetinandcurcuminbionanovesiclesforthepreventionand rapidregenerationoffull-thicknessskindefectsonmice.ActaBiomater 2014;10:1292–300.
[11]AlaseirlisDA,LiY,CilliF,FuFH,WangJH-C.Decreasinginflammatoryresponse ofinjuredpatellartendonsresultsinincreasedcollagenfibrildiameters. ConnectTissueRes2005;46:12–7.
[12]ParkHB,HahY-S,YangJ-W,NamJ-B,ChoS-H,JeongS-T.Antiapoptoticeffects ofanthocyaninsonrotatorcufftenofibroblasts.JOrthopRes2010;28:1162–9.
[13]GüleçA,TürkY,AydinBK,ErkoçakÖF,SafalıS,UgurluogluC.Effectofcurcumin ontendonhealing:anexperimentalstudyinaratmodelofAchillestendon injury.IntOrthop2018;42:1905–10.
[14]NamD, Mau E,WangY, Wright D, SilkstoneD, Whetstone H,etal. T-lymphocytesenableosteoblastmaturationviaIL-17Fduringtheearlyphaseof fracturerepair.PLoSOne2012;7:e40044.
[15]EdderkaouiB.Potentialroleofchemokinesinfracturerepair.FrontEndocrinol 2017;8:39.
[16]PoniatowskiŁA,WojdasiewiczP,GasikR,SzukiewiczD.Transforminggrowth factorbetafamily:insightintotheroleofgrowthfactorsinregulationof fracturehealingbiologyandpotentialclinicalapplications.MediatorsInflamm 2015;2015:1–17.
[17]Aydin K, Sahin V, Gürsu S, Mercan AS, Demir B, Yildirim T. Effect of pentoxifyllineonfracturehealing:anexperimentalstudy.EklemHastalik Cerrahisi2011;22:160–5.
[18]LiG,ChenL,ChenK.Curcuminpromotesfemoralfracturehealinginarat modelbyactivationofautophagy.MedSciMonit2018;24:4064–72. [19]SajithlalGB,ChithraP,ChandrakasanG.Effectofcurcuminontheadvanced
glycationandcross-linkingofcollagenindiabeticrats.BiochemPharmacol 1998;56:1607–14.
[20]HuoMH,TroianoNW,PelkerRR,GundbergCM,FriedlaenderGE.Theinfluence ofibuprofenonfracturerepair:biomechanical,biochemical,histologic,and histomorphometricparametersinrats.JOrthopRes1991;9:383–90. [21]FolwarcznaJ,ZychM,TrzeciakHI.Effectsofcurcuminontheskeletalsystemin
rats.PharmacolRep2010;62:900–9.