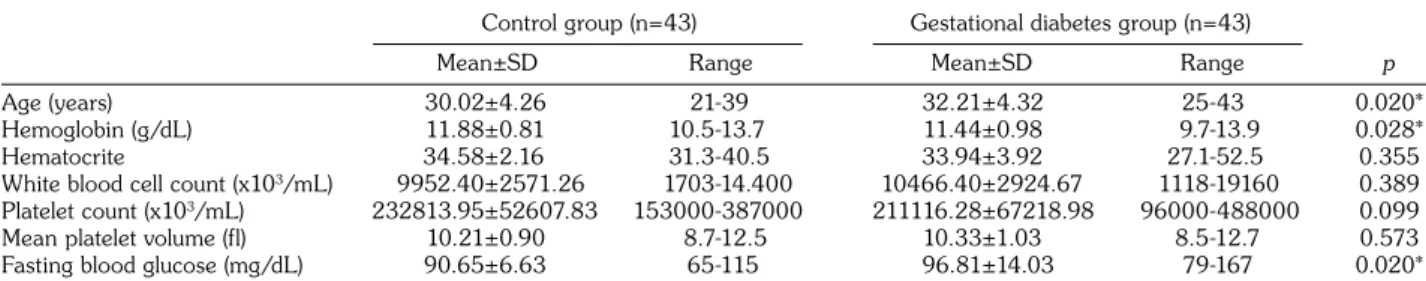
Mean platelet volume in gestational diabetes mellitus
Tam metin
Şekil
Benzer Belgeler
The main findings of our study was that compared with the patients with normal renal arteries or non-critical ARAS, the patients with critical ARAS
They assessed the relationship between mean platelet volume (MPV), platelet count, platelet hematocrit and high on-treatment platelet reac- tivity (HTPR) in patients with
No significant difference was found between the patient group with acromegaly and the healthy control group in terms of peripheral systolic blood pressure, peripheral main arterial
Although there are no statistically difference between three groups in terms of diabetes mellitus, hypertension and smoking, they did not mention about the body mass
Entrepreneurial success or failure depends on the accessibility of financial resources like debt, equity, grants and subsidies, the degree to which government policies
The main reason why political values meet the requirements of the development of our society, the vital needs of the state independence of Uzbekistan, the interests of our people
In conclusion, significantly higher MPV in patients with HcV infection may be responsible for the viral effect that causes platelet dysfunction, supporting the association between
The aim of the present study was to show the NLR and MPV values in patients who have COM with or without ossicular/bony destruction or cholesteatoma, to compare the results