Monodisperse Pt-Co/GO anodes with varying Pt: Co ratios as highly active and stable electrocatalysts for methanol electrooxidation reaction
Tam metin
Şekil

Benzer Belgeler
Yine örneklemdeki kişilerin narsisizm düzeyi ile Instagram, Facebook, Twitter, WhatsApp, LinkledIn, Swarm ve Snapchat gibi sosyal medya araçları kullanım
Although the name of chapter 7 is prime ideals, our main object there is finding the primitive idempotents of the monomial Burnside rings tensored over Z with an integral domain
According to this there are three departments that are responsible from the purchasing operations: ‘Parça Temin Müdürlüğü’ which carries out the purchasing
Ampirik analizin sonuçlarına göre performans beklentisi ve sosyal etki faktörlerinin kullanım niyeti üzerinde olumlu bir etkiye sahip olduğu, fakat çaba
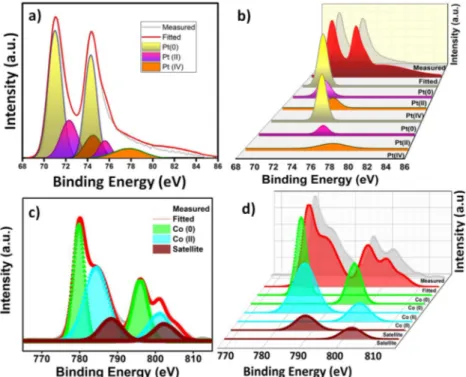
ġen ve arkadaĢları, indirgenmiĢ grafen oksit destekli platin (Pt@rGO) heterojen katalizör varlığında, çevre dostu hidrojen kaynağı dimetilamin boran (DMAB) çözücü
Ka‘b, bu meclisin ihtişamına işaret ederek, “Öyle bir meclise hazır oldum ki, öyle muazzam şeyler işittim ve gördüm ki, şayet fil bunlara vakıf olsaydı
Gruplara göre öğrencilerin görüĢlerinin dağılımına baktığımızda 15 öğrenci “hiç duymadığını”; 1 öğrenci “az duyduğunu”; 4 öğrencinin “çevrenin
Architects and designers can plug applica- tion-level data structures and functions into this template to generate hardware imple- mentations for a large class of graph