The role of hemogram parameters and CRP in predicting mortality in COVID‐19 infection
Tam metin
Şekil


Benzer Belgeler
Demographic and clinical data such as age, gender, vascular risk factors, antithrombotic or anticoagulant use, presence of symptomatic cSAH or intracerebral
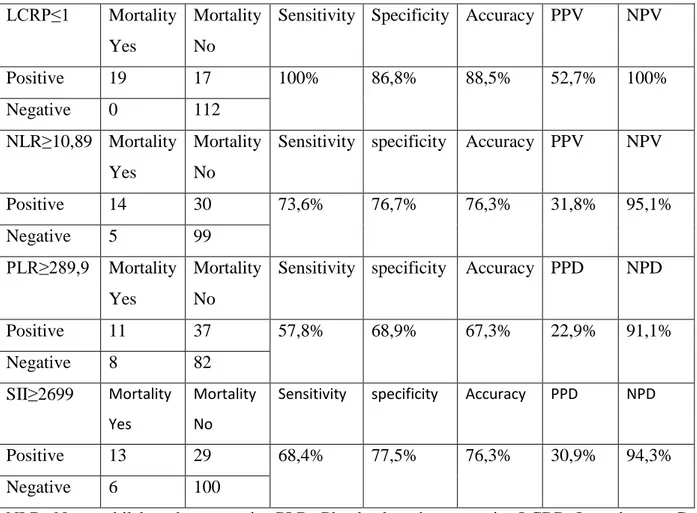
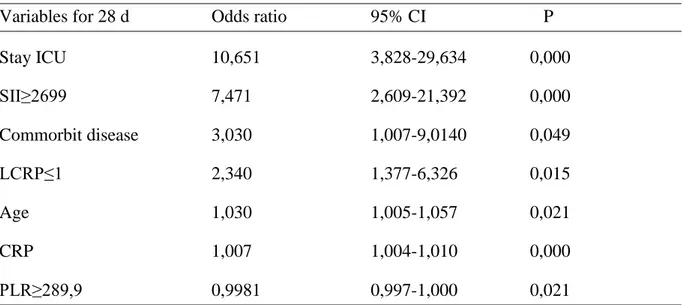
Objective: To investigate the association of white blood cell (WBC) counts, neutrophil, platelets, lymphocyte counts, C-reactive protein (CRP), neutrophil / lymphocyte ratio
Currently, some authors have assessed the predictive and prog- nostic role of hematological parameters in prostate cancer using the neutrophil-to-lymphocyte ratio, mean
Age, gender, type of trauma, GCS, Revised Trauma Score (RTS), Abbreviated Injury Scale (AIS) and Injury Severity Score (ISS) values on admission, requirement
On March 11, 2020, It is declared as a pandemic by the World Health Organization and within the same day, the first case o the new Coronavirus Disease-2019 (COVID-19) in Turkey
In Velisaris et al.’s 8 study investigating the NLR and sepsis severity scoring systems at admission, the data of 50 patients with an average age of 68.4 years were
Comparison of D-dimer Level Measured on the Third Day of Hospitalization with Admission D-dimer Level in Predicting In-hospital Mortality in COVID-19 Patients.. COVID-19 Hastalarında
Objective: The present study aimed to compare the value of D-dimer measured on the 3rd day of hospitaliza- tion with admission D-dimer level in predicting in-hospital mortality